Chemical Physics Letters ( IF 2.8 ) Pub Date : 2020-10-06 , DOI: 10.1016/j.cplett.2020.138059 Mohan Tiwari , C.N. Ramachandran
|
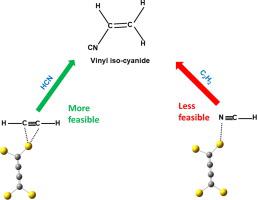
The reaction between hydrogen cyanide and acetylene over carbon intercalated gold clusters forming the industrially important product vinyl iso-cyanide (H2C = CHNC) is investigated using density functional methods. The free energy of desorption of the product vinyl iso-cyanide was found to be significantly less than the energy barrier for the reaction suggesting that the catalysts are not poisoned after the conversion. The reaction is more feasible when it is initiated by acetylene than hydrogen cyanide. The pre-adsorption of reactants at multiple sites of C4Au4 is examined and the co-operativity and site specificity of the reaction are discussed.
中文翻译:

碳插层金簇上乙炔的氢氰化:协同性和位点特异性
使用密度泛函方法研究了氰化氢与乙炔在碳插层金簇上形成工业上重要的产物乙烯基异氰(H 2 C = CHNC)的反应。发现产物乙烯基异氰化物的解吸自由能明显小于反应的能垒,表明催化剂在转化后没有被中毒。由乙炔引发的反应比氰化氢更可行。检查了反应物在C 4 Au 4多个位置上的预吸附,并讨论了反应的协同性和位置特异性。



























 京公网安备 11010802027423号
京公网安备 11010802027423号