当前位置:
X-MOL 学术
›
Chem. Heterocycl. Comp.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
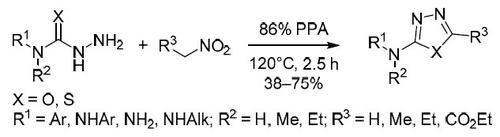
Preparation of 1,3,4-oxadiazoles and 1,3,4-thiadiazoles via chemoselective сyclocondensation of electrophilically activated nitroalkanes to (thio)semicarbazides or thiohydrazides
Chemistry of Heterocyclic Compounds ( IF 1.5 ) Pub Date : 2020-09-24 , DOI: 10.1007/s10593-020-02775-5 Nicolai A. Aksenov , Nikolai A. Arutiunov , Nikita K. Kirillov , Dmitrii A. Aksenov , Alexander V. Aksenov , Michael Rubin
中文翻译:

通过亲电活化的硝基烷烃的化学选择性缩合为(硫代)氨基脲或硫酰肼制备1,3,4-恶二唑和1,3,4-噻二唑
更新日期:2020-09-24
Chemistry of Heterocyclic Compounds ( IF 1.5 ) Pub Date : 2020-09-24 , DOI: 10.1007/s10593-020-02775-5 Nicolai A. Aksenov , Nikolai A. Arutiunov , Nikita K. Kirillov , Dmitrii A. Aksenov , Alexander V. Aksenov , Michael Rubin
|
Unusual reaction proceeding via the electrophilic activation of nitroalkanes in the presence of polyphosphoric acid has been discovered. Subsequent nucleophilic attack with semicarbazides or thiosemicarbazides allows to access 2-amino-1,3,4-oxadiazoles and 2-amino-1,3,4-thiadiazoles, respectively.
中文翻译:

通过亲电活化的硝基烷烃的化学选择性缩合为(硫代)氨基脲或硫酰肼制备1,3,4-恶二唑和1,3,4-噻二唑
已经发现在多磷酸存在下经由硝基链烷的亲电活化进行的异常反应。随后用氨基脲或硫代氨基脲的亲核攻击使得可以分别接近2-氨基-1,3,4-恶二唑和2-氨基-1,3,4-噻二唑。


























 京公网安备 11010802027423号
京公网安备 11010802027423号