Chemical Physics Letters ( IF 2.8 ) Pub Date : 2020-09-22 , DOI: 10.1016/j.cplett.2020.138025 Luyi Ji , Yongtao Wang , Liangliang Huang , Yingchun Liu , Qi Wang
|
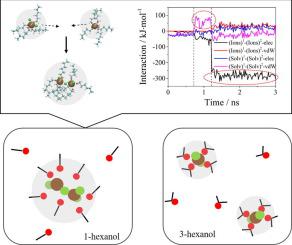
Copper ion is a common catalyst in the industrial process, where solvents are known to affect its catalytic efficiency. In this work, we studied the form and distribution of CuCl2 clusters in 1-hexanol and 3-hexanol for 2,3,6-trimethylphenol oxidation. Our results reveal that copper aggregation occurs in both solvents under relatively high CuCl2 concentration. But in low concentration, aggregation only exists in 1-hexanol. Besides, the aggregation is dominated by the competition between Cu-Cl electrostatic and van der Waals interactions of the solvents. Our results may provide a fundamental insight for understanding the product selectivity of phenol oxidation in hexanol solutions.
中文翻译:

1-己醇和3-己醇溶液中氯化铜聚集体的协同相互作用用于多核催化氧化
铜离子是工业过程中常见的催化剂,已知溶剂会影响其催化效率。在这项工作中,我们研究了1,2,3,6-三甲基苯酚氧化过程中1-己醇和3-己醇中CuCl 2团簇的形成和分布。我们的结果表明,在相对较高的CuCl 2浓度下,两种溶剂中都会发生铜聚集。但是在低浓度下,聚集仅存在于1-己醇中。此外,聚集主要由Cu-Cl静电和溶剂的范德华相互作用之间的竞争所决定。我们的结果可能为了解己醇溶液中苯酚氧化的产物选择性提供基本的见识。



























 京公网安备 11010802027423号
京公网安备 11010802027423号