Bioorganic Chemistry ( IF 5.1 ) Pub Date : 2020-09-19 , DOI: 10.1016/j.bioorg.2020.104293 Daiki Kaneko , Masayuki Ninomiya , Rina Yoshikawa , Yukari Ono , Amol D. Sonawane , Kaori Tanaka , Atsuyoshi Nishina , Mamoru Koketsu
|
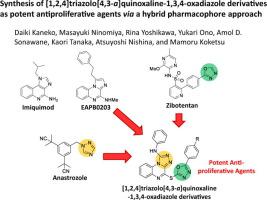
Imiquimod (1-isobutyl-1H-imidazo[4,5-c]quinolin-4-amine) is efficacious in topical therapy for certain types of skin cancers. Structurally similar EAPB0203 (N-methyl-1-(2-phenethyl)imidazo[1,2-a]quinoxalin-4-amine) has been shown higher in vitro potency than imiquimod. Besides, triazole, oxadiazole, and thiadiazole rings are privileged building blocks in drug design. A series of [1,2,4]triazolo[4,3-a]quinoxaline-1,3,4-oxadiazole and [1,2,4]triazolo[4,3-a]quinoxaline-1,3,4-thiadiazole derivatives were therefore synthesized by incorporation of these rings into the structure of EAPB0203 and assessed their antiproliferative effects against various cancer cell lines. The 1,3,4-oxadiazole derivatives demonstrated the superior effectiveness compared to imiquimod and EAPB0203. Our findings highlight the excellent potential of [1,2,4]triazolo[4,3-a]quinoxaline-1,3,4-oxadiazole derivatives as anticancer agents.
中文翻译:

通过混合药效团方法合成[1,2,4]三唑并[4,3 - a ]喹喔啉-1,3,4-恶二唑衍生物作为有效的抗增殖剂
咪喹莫特(1-异丁基-1 H-咪唑并[4,5 - c ]喹啉-4-胺)对某些类型的皮肤癌有效。结构相似的EAPB0203(N-甲基-1-(2-苯乙基)咪唑并[1,2 - a ]喹喔啉-4-胺)的体外药效比咪喹莫特更高。此外,三唑,恶二唑和噻二唑环是药物设计中的重要组成部分。一系列[1,2,4]三唑并[4,3 - a ]喹喔啉-1,3,4-恶二唑和[1,2,4]三唑并[4,3- a因此,通过将这些环并入EAPB0203的结构中来合成]喹喔啉-1,3,4-噻二唑衍生物,并评估它们对多种癌细胞系的抗增殖作用。与咪喹莫特和EAPB0203相比,1,3,4-恶二唑衍生物表现出卓越的功效。我们的发现突出了[1,2,4]三唑并[4,3 - a ]喹喔啉-1,3,4-恶二唑衍生物作为抗癌剂的巨大潜力。


























 京公网安备 11010802027423号
京公网安备 11010802027423号