Bioorganic Chemistry ( IF 5.1 ) Pub Date : 2020-09-17 , DOI: 10.1016/j.bioorg.2020.104278 Daniela de Luna Martins , Adriel Alves Borges , Nayane A. do A. e Silva , Juliana Vieira Faria , Lucas Villas Bôas Hoelz , Hellen Valério Chaves Moura de Souza , Murilo Lamim Bello , Nubia Boechat , Vitor Francisco Ferreira , Robson Xavier Faria
|
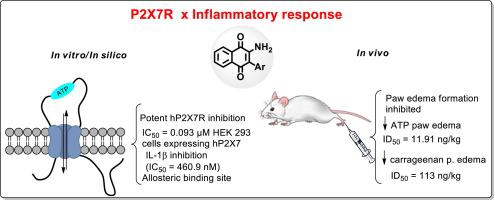
Extracellular ATP activates purinergic receptors such as P2X7, cationic channels for Ca2+, K+, and Na+. There is robust evidence of the involvement of these receptors in the immune response, so P2X7 receptors (P2X7R) are considered a potential therapeutic target for the development of anti-inflammatory drugs. Although there are many studies of the anti-inflammatory properties of naphthoquinones, these molecules have not yet been explored as P2X7 antagonists. In previous work, our group prepared 3-substituted (halogen or aryl) 2-hydroxy-1,4-naphthoquinones and studied their action on P2X7R. In this paper, eight 2-amino-3-aryl-1,4-naphthoquinones were evaluated to identify the inhibitory activity on P2X7R and the toxicological profile. Three analogues (AD-4CN, AD-4Me, and AD-4F) exhibited reduced toxicity for mammalian cells with CC50 values higher than 500 µM. These three 3-substituted 2-amino-1,4-naphthoquinones inhibited murine P2X7R (mP2X7R) in vitro. However, the analogues AD-4CN and AD-4Me showed low selectivity index values. AD-4F inhibited both mP2X7R and human P2X7R (hP2X7R) with IC50 values of 0.123 and 0.93 µM, respectively. Additionally, this analogue exhibited higher potency than BBG at inhibiting the ATP-induced release of IL-1β in vitro. Carrageenan-induced paw edema in vivo was reversed for AD-4F with an ID50 value of 11.51 ng/kg. Although AD-4F was less potent than previous 3-substituted (halogen or aryl) 2-hydroxy-1,4-naphthoquinones such as AN-04 in vitro, this 3-substituted 2-amino-1,4-naphthoquinone revealed higher potency in vivo to reduce the edematogenic response. In silico analysis suggests that the binding site of the novel 2-amino-3-aryl-1,4-naphthoquinone derivatives, including all the tautomeric forms, is located in the pore area of the hP2X7R model. Based on these results, we considered AD-4F to be a satisfactory P2X7R inhibitor. AD-4F might be used as a scaffold structure to design a novel series of inhibitors with potential inhibitory activity on murine (mP2X7R) and human (hP2X7R) P2X7 receptors.
中文翻译:

P2X7受体被2-氨基-3-芳基-1,4-萘醌抑制
细胞外ATP激活嘌呤能受体,例如P2X7,Ca 2 +,K +和Na +的阳离子通道。有强有力的证据表明这些受体参与了免疫反应,因此P2X7受体(P2X7R)被认为是抗炎药开发的潜在治疗靶标。尽管已经对萘醌的抗炎特性进行了许多研究,但尚未探索这些分子作为P2X7拮抗剂。在以前的工作中,我们的小组准备了3-取代的(卤素或芳基)2-羟基-1,4-萘醌,并研究了它们对P2X7R的作用。在本文中,评估了八个2-氨基-3-芳基-1,4-萘醌,以确定其对P2X7R的抑制活性和毒理学特征。三种类似物(AD-4CN,AD-4Me和AD-4F)对CC 50值高于500 µM的哺乳动物细胞毒性降低。这三个3-取代的2-氨基-1,4-萘醌在体外抑制鼠P2X7R(mP2X7R)。然而,类似物AD-4CN和AD-4Me显示出低的选择性指数值。AD-4F抑制mP2X7R和人P2X7R(hP2X7R),IC 50值分别为0.123和0.93 µM。另外,该类似物在体外抑制ATP诱导的IL-1β释放方面显示出比BBG更高的效力。角叉菜胶在体内引起的爪水肿被逆转AD-4F的ID 50值为11.51 ng / kg。尽管在体外AD-4F的效力不及先前的3-取代的(卤素或芳基)2-羟基-1,4-萘醌,例如AN-04 ,但这种3-取代的2-氨基-1,4-萘醌显示出更高的效力体内减少水肿反应。电脑分析表明,新型2-氨基-3-芳基-1,4-萘醌衍生物(包括所有互变异构形式)的结合位点位于hP2X7R模型的孔区域。根据这些结果,我们认为AD-4F是令人满意的P2X7R抑制剂。AD-4F 可以用作支架结构来设计一系列对鼠(mP2X7R)和人(hP2X7R)P2X7受体具有潜在抑制活性的抑制剂。

























 京公网安备 11010802027423号
京公网安备 11010802027423号