Separation and Purification Technology ( IF 8.6 ) Pub Date : 2020-09-15 , DOI: 10.1016/j.seppur.2020.117633 Zhenzhen Huang , Yuanhang Wei , Zhongxian Song , Jiawen Luo , Yanli Mao , Jingqing Gao , Xuejun Zhang , Can Niu , Haiyan Kang , Zhaodong Wang
|
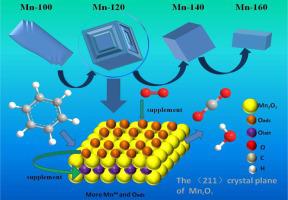
A series of Mn2O3 catalysts were prepared by different hydrothermal temperatures and employed to the catalytic oxidation of benzene due to their high activity. The Mn-120 sample with three-dimensional (3D) hierarchical cube-like morphology mainly exposed (2 1 1) crystal plane exhibited the best catalytic activity for the oxidation of benzene and achieved the benzene conversion of 90% at 248 °C. Meanwhile, the best catalytic performance of the Mn-120 sample was attributed to the most amounts of the Mn3+ species and surface-adsorbed oxygen. With the increase of Mn3+ species, the oxygen binding capacity of Mn-120 was reduced, which could increase its oxygen mobility. The abundant adsorbed oxygen species of Mn-120 could improve the replenishment capacity of lattice oxygen species, and the reaction pathway of catalytic oxidation of benzene over MnO2 obeyed the Mars-Van-Krevelen (MVK) mechanism.
中文翻译:

高效净化苯燃烧的三维(3D)分级Mn 2 O 3催化剂
在不同的水热温度下制备了一系列Mn 2 O 3催化剂,并由于其高活性而用于苯的催化氧化。主要暴露于(2 1 1)晶面的具有三维(3D)分层立方状形态的Mn-120样品表现出对苯氧化的最佳催化活性,并在248°C时实现了90%的苯转化率。同时,Mn-120样品的最佳催化性能归因于大量的Mn 3+种类和表面吸附的氧气。随着Mn 3+的增加物种中,Mn-120的氧结合能力降低,这可能增加其氧迁移率。Mn-120的大量吸附氧可以提高晶格氧的补充能力,苯在MnO 2上催化氧化的反应途径遵循Mars-Van-Krevelen(MVK)机理。


























 京公网安备 11010802027423号
京公网安备 11010802027423号