Bioorganic & Medicinal Chemistry Letters ( IF 2.7 ) Pub Date : 2020-09-11 , DOI: 10.1016/j.bmcl.2020.127547 Yoshiyuki Taoda 1 , Masayoshi Miyagawa 1 , Toshiyuki Akiyama 1 , Kenji Tomita 1 , Yasushi Hasegawa 1 , Ryu Yoshida 1 , Takeshi Noshi 1 , Takao Shishido 1 , Makoto Kawai 1
|
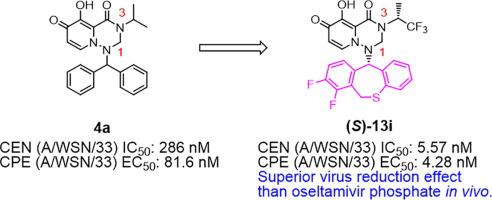
This work describes a set of discovery research studies of an influenza cap-dependent endonuclease (CEN) inhibitor with a carbamoyl pyridone bicycle (CAB) scaffold. Using influenza CEN inhibitory activity, antiviral activity and pharmacokinetic (PK) parameters as indices, structure activity relationships (SAR) studies were performed at the N-1 and N-3 positions on the CAB scaffold, which is a unique template to bind two metals. The hydrophobic substituent at the N-1 position is extremely important for CEN inhibitory activity and antiviral activity, and dihydrodibenzothiepine is the most promising pharmacophore. The compound (S)-13i showed potent virus titer reduction over oseltamivir phosphate in an in vivo mouse model. The CAB compound described herein served as the lead compound of baloxavir marboxil with a tricyclic scaffold, which was approved in Japan and the USA in 2018.
中文翻译:

Dihydrodibenzothiepine:流行性感冒帽依赖性核酸内切酶抑制剂中有望的疏水药效基团。
这项工作描述了一组与氨基甲酰吡啶酮自行车(CAB)支架结合的流感帽依赖性核酸内切酶(CEN)抑制剂的发现研究。使用流感CEN抑制活性,抗病毒活性和药代动力学(PK)参数作为指标,在CAB支架的N-1和N-3位置进行了结构活性关系(SAR)研究,这是结合两种金属的独特模板。N-1位的疏水取代基对于CEN抑制活性和抗病毒活性极为重要,而二氢二苯并噻吩平是最有前途的药效团。化合物(S)-13i在体内显示出比磷酸奥司他韦有效的病毒效价降低鼠标模型。本文所述的CAB化合物用作具有三环支架的baloxavir marboxil的前导化合物,该化合物已于2018年在日本和美国获批。

























 京公网安备 11010802027423号
京公网安备 11010802027423号