当前位置:
X-MOL 学术
›
Organometallics
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Probing for Four-Coordinate Zerovalent Iron in a π-Acidic Ligand Field: A Functional Source of FeL4 Enabled by Labile Dinitrogen Binding
Organometallics ( IF 2.8 ) Pub Date : 2020-09-10 , DOI: 10.1021/acs.organomet.0c00487 Myles J. Drance 1 , Shuai Wang 1 , Milan Gembicky 1 , Arnold L. Rheingold 1 , Joshua S. Figueroa 1
Organometallics ( IF 2.8 ) Pub Date : 2020-09-10 , DOI: 10.1021/acs.organomet.0c00487 Myles J. Drance 1 , Shuai Wang 1 , Milan Gembicky 1 , Arnold L. Rheingold 1 , Joshua S. Figueroa 1
Affiliation
|
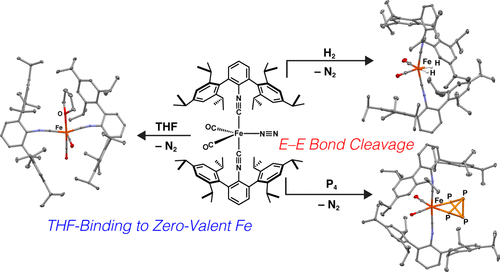
It has long been recognized that the first intermediate in chemical reactions mediated by Fe(CO)5 is Fe(CO)4. However, the extreme instability of this unsaturated, high-spin species has hampered detailed structural and stoichiometric reactivity studies. The previously reported heteroleptic complex Fe(N2)(CO)2(CNArTripp2)2 (ArTripp2 = 2,6-(2,4,6-(i-Pr)3C6H2)2C6H3), which is isolobal to Fe(CO)5, is shown here to possess a labile dinitrogen ligand that allows it to perform reactions analogous to Fe(CO)4. Specifically, Fe(N2)(CO)2(CNArTripp2)2 oxidatively adds dihydrogen, white phosphorus, and the Si–H bonds of triethylsilane and phenylsilane readily at room temperature and also binds tetrahydrofuran in the absence of N2. Accordingly, Fe(N2)(CO)2(CNArTripp2)2 effectively serves as a masked form of Fe(CO)4, allowing for well-defined reactivity and structurally characterizable reaction products.
中文翻译:

π酸配体场中四坐标零价铁的探测:通过不稳定的二氮键结合实现的FeL 4的功能来源
早就认识到,由Fe(CO)5介导的化学反应中的第一中间体是Fe(CO)4。但是,这种不饱和的高自旋物种的极度不稳定性阻碍了详细的结构和化学计量反应性研究。先前报道的杂合配合物Fe(N 2)(CO)2(CNAr Tripp2)2(Ar Tripp2 = 2,6-(2,4,6-(i -Pr)3 C 6 H 2)2 C 6 H 3),与Fe(CO)5等价此处显示的,具有不稳定的二氮配体,可使其进行类似于Fe(CO)4的反应。特别是,Fe(N 2)(CO)2(CNAr Tripp2)2在室温下容易氧化氧化二氢,白磷以及三乙基硅烷和苯基硅烷的Si-H键,并且在不存在N 2的情况下与四氢呋喃结合。因此,Fe(N 2)(CO)2(CNAr Tripp2)2有效地充当了Fe(CO)4的掩蔽形式,允许明确的反应性和结构可表征的反应产物。
更新日期:2020-09-28
中文翻译:

π酸配体场中四坐标零价铁的探测:通过不稳定的二氮键结合实现的FeL 4的功能来源
早就认识到,由Fe(CO)5介导的化学反应中的第一中间体是Fe(CO)4。但是,这种不饱和的高自旋物种的极度不稳定性阻碍了详细的结构和化学计量反应性研究。先前报道的杂合配合物Fe(N 2)(CO)2(CNAr Tripp2)2(Ar Tripp2 = 2,6-(2,4,6-(i -Pr)3 C 6 H 2)2 C 6 H 3),与Fe(CO)5等价此处显示的,具有不稳定的二氮配体,可使其进行类似于Fe(CO)4的反应。特别是,Fe(N 2)(CO)2(CNAr Tripp2)2在室温下容易氧化氧化二氢,白磷以及三乙基硅烷和苯基硅烷的Si-H键,并且在不存在N 2的情况下与四氢呋喃结合。因此,Fe(N 2)(CO)2(CNAr Tripp2)2有效地充当了Fe(CO)4的掩蔽形式,允许明确的反应性和结构可表征的反应产物。

























 京公网安备 11010802027423号
京公网安备 11010802027423号