当前位置:
X-MOL 学术
›
Tetrahedron
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Synthesis of glycosides of 1H-Pyrazolo[3,4-b]pyridin-3(2H)-ones
Tetrahedron ( IF 2.1 ) Pub Date : 2020-09-07 , DOI: 10.1016/j.tet.2020.131522 Linda Supe , Martin Hein , Viktor O. Iaroshenko , Alexander Villinger , Peter Langer
中文翻译:

1 H-吡唑并[3,4- b ]吡啶-3(2 H)-ones苷的合成
更新日期:2020-10-12
Tetrahedron ( IF 2.1 ) Pub Date : 2020-09-07 , DOI: 10.1016/j.tet.2020.131522 Linda Supe , Martin Hein , Viktor O. Iaroshenko , Alexander Villinger , Peter Langer
|
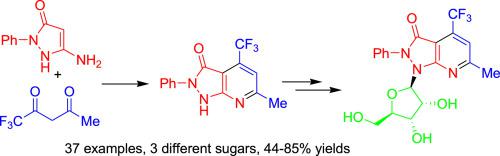
A number of new fluorinated and non-fluorinated glycosides of 1H-pyrazolo[3,4-b]pyridin-3(2H)-ones were synthesized by direct attachment of the carbohydrate moiety to the heterocycle using the silyl Hilbert-Jones glycosylation method. The products were obtained in good to excellent yields and with very good anomeric stereoselectivity. The starting 1H-pyrazolo[3,4-b]pyridin-3(2H)-ones were prepared by regioselective cyclization of 1,3-dicarbonyl compounds with electron-rich heterocycles containing an enamine functionality.
中文翻译:

1 H-吡唑并[3,4- b ]吡啶-3(2 H)-ones苷的合成
通过使用甲硅烷基希尔伯特-琼斯糖基化将碳水化合物部分直接连接到杂环上,合成了许多1 H-吡唑并[3,4- b ]吡啶3(2 H)-的新的氟化和非氟化糖苷方法。获得的产物具有良好的产率至优异的产率以及非常好的异头立体选择性。起始的1 H-吡唑并[3,4- b ]吡啶-3(2 H)-是通过1,3-二羰基化合物与含有烯胺官能团的富电子杂环的区域选择性环化制备的。



























 京公网安备 11010802027423号
京公网安备 11010802027423号