当前位置:
X-MOL 学术
›
J. Chem. Eng. Data
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
CO2, N2, and H2 Adsorption by Hyper-Cross-Linked Polymers and Their Selectivity Evaluation by Gas–Solid Equilibrium
Journal of Chemical & Engineering Data ( IF 2.6 ) Pub Date : 2020-09-01 , DOI: 10.1021/acs.jced.0c00541 Hamid Ramezanipour Penchah 1 , Hossein Ghanadzadeh Gilani 1 , Ahad Ghaemi 2
Journal of Chemical & Engineering Data ( IF 2.6 ) Pub Date : 2020-09-01 , DOI: 10.1021/acs.jced.0c00541 Hamid Ramezanipour Penchah 1 , Hossein Ghanadzadeh Gilani 1 , Ahad Ghaemi 2
Affiliation
|
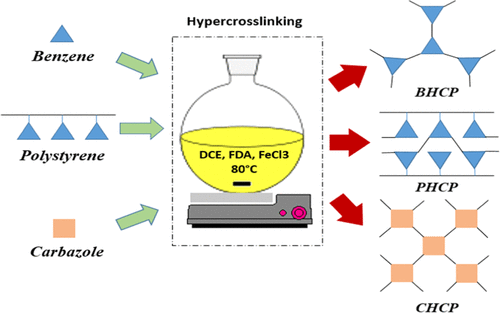
In this study, the effect of hyper-cross-linked adsorbent structural parameters on gas adsorption and selectivity was investigated. Hyper-cross-linked polymere (HCP) adsorbents were synthesized based on three widely used monomers, such as benzene, polystyrene, and carbazole. Cross-linker value and synthesis time were considered as structural parameters. The selectivities of CO2/N2 and CO2/H2 at 1 bar and 298 K were evaluated by the ideal adsorption solution theory (IAST) method. The results showed that carbazole-based HCP adsorbents had higher selectivity of CO2/N2 and CO2/H2 than benzene- and polystyrene-based HCPs, and also benzene-based HCPs had higher adsorption capacity. In all three adsorbents, selectivity increases with increasing synthesis time at a constant cross-linker value. In benzene- and carbazole-based adsorbents, selectivity increases with increasing cross-linker value in constant synthesis time, while in polystyrene-based adsorbents, it decreases. Finally, the real adsorption solution theory (RAST) and activity coefficient in the gas–solid equilibrium was applied due to the difference between real and computational mole fractions in the adsorbed phase. The activity coefficient constants were calculated by genetic algorithm and minimization of difference between experimental and calculated data. The results show that the calculated data with the RAST method have a good agreement with experimental data.
中文翻译:

超交联聚合物对CO 2,N 2和H 2的吸附及其气固平衡的选择性评估
在这项研究中,研究了超交联的吸附剂结构参数对气体吸附和选择性的影响。基于三种广泛使用的单体(如苯,聚苯乙烯和咔唑)合成了超交联聚合物(HCP)吸附剂。交联剂值和合成时间被认为是结构参数。通过理想吸附溶液理论(IAST)方法评估了1 bar和298 K时CO 2 / N 2和CO 2 / H 2的选择性。结果表明,咔唑类HCP吸附剂具有较高的CO 2 / N 2和CO 2 / H 2选择性。与苯和聚苯乙烯基六氯苯酚相比,苯基六氯苯酚具有更高的吸附能力。在所有三种吸附剂中,在恒定的交联剂值下,选择性随合成时间的增加而增加。在苯和咔唑类吸附剂中,在恒定的合成时间内,选择性随交联剂值的增加而增加,而在聚苯乙烯类吸附剂中,选择性则降低。最后,由于吸附相中实际摩尔分数和计算摩尔分数之间存在差异,因此应用了实际吸附溶液理论(RAST)和气固平衡中的活度系数。通过遗传算法计算活性系数常数,并最小化实验数据与计算数据之间的差异。结果表明,RAST法计算的数据与实验数据吻合良好。
更新日期:2020-10-08
中文翻译:

超交联聚合物对CO 2,N 2和H 2的吸附及其气固平衡的选择性评估
在这项研究中,研究了超交联的吸附剂结构参数对气体吸附和选择性的影响。基于三种广泛使用的单体(如苯,聚苯乙烯和咔唑)合成了超交联聚合物(HCP)吸附剂。交联剂值和合成时间被认为是结构参数。通过理想吸附溶液理论(IAST)方法评估了1 bar和298 K时CO 2 / N 2和CO 2 / H 2的选择性。结果表明,咔唑类HCP吸附剂具有较高的CO 2 / N 2和CO 2 / H 2选择性。与苯和聚苯乙烯基六氯苯酚相比,苯基六氯苯酚具有更高的吸附能力。在所有三种吸附剂中,在恒定的交联剂值下,选择性随合成时间的增加而增加。在苯和咔唑类吸附剂中,在恒定的合成时间内,选择性随交联剂值的增加而增加,而在聚苯乙烯类吸附剂中,选择性则降低。最后,由于吸附相中实际摩尔分数和计算摩尔分数之间存在差异,因此应用了实际吸附溶液理论(RAST)和气固平衡中的活度系数。通过遗传算法计算活性系数常数,并最小化实验数据与计算数据之间的差异。结果表明,RAST法计算的数据与实验数据吻合良好。

























 京公网安备 11010802027423号
京公网安备 11010802027423号