当前位置:
X-MOL 学术
›
Adv. Synth. Catal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Functionalization of the Imidazole Backbone by Means of a Tailored and Optimized Oxidative Heck Cross‐Coupling
Advanced Synthesis & Catalysis ( IF 5.4 ) Pub Date : 2020-08-25 , DOI: 10.1002/adsc.202000909 Davide Cirillo 1 , Francesco Angelucci 1 , Hans‐René Bjørsvik 1
Advanced Synthesis & Catalysis ( IF 5.4 ) Pub Date : 2020-08-25 , DOI: 10.1002/adsc.202000909 Davide Cirillo 1 , Francesco Angelucci 1 , Hans‐René Bjørsvik 1
Affiliation
|
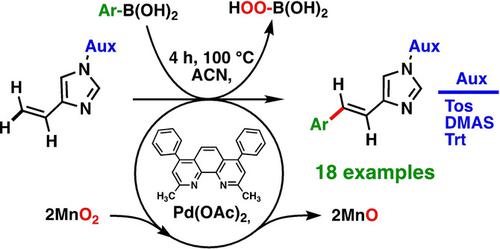
A general and selective Pd‐catalyzed cross‐coupling of aromatic boronic acids with vinyl‐imidazoles is disclosed. Unlike most cross‐coupling reactions, this method operates well in absence of bases avoiding the formation of by‐products. The reactivity is highly enhanced by the presence of nitrogen‐based ligands, in particular bathocuproine. The method involves MnO2 as oxidant for the oxidation Pd (0)→Pd (II), a much weaker oxidant than previously reported in the literature. This allows for the use of reactants that possess a multitude of functional groups. A scope and limitation study involving a series of 24 boronic acids, whereof 18 afforded TMs in yields in the range 41–95%. The disclosed method constitutes the first general method for the oxidative Heck cross‐coupling on the imidazole scaffold, which moreover operates with a selection of other heterocycles.
中文翻译:

通过量身定制和优化的氧化Heck交叉偶联功能实现咪唑骨架的功能化
公开了芳族硼酸与乙烯基咪唑的一般和选择性Pd催化交叉偶联。与大多数交叉偶联反应不同,该方法在没有碱的情况下运行良好,避免了副产物的形成。氮基配体(特别是红霉素)的存在可大大提高反应性。该方法涉及MnO 2作为氧化Pd(0)→Pd(II)的氧化剂,它比以前文献中报道的要弱得多。这允许使用具有多个官能团的反应物。范围和限度研究涉及一系列24种硼酸,其中18种提供的TM产率在41–95%范围内。所公开的方法构成了在咪唑支架上进行氧化性Heck交叉偶联的第一种通用方法,该方法还可以与其他杂环一起选择。
更新日期:2020-08-25
中文翻译:

通过量身定制和优化的氧化Heck交叉偶联功能实现咪唑骨架的功能化
公开了芳族硼酸与乙烯基咪唑的一般和选择性Pd催化交叉偶联。与大多数交叉偶联反应不同,该方法在没有碱的情况下运行良好,避免了副产物的形成。氮基配体(特别是红霉素)的存在可大大提高反应性。该方法涉及MnO 2作为氧化Pd(0)→Pd(II)的氧化剂,它比以前文献中报道的要弱得多。这允许使用具有多个官能团的反应物。范围和限度研究涉及一系列24种硼酸,其中18种提供的TM产率在41–95%范围内。所公开的方法构成了在咪唑支架上进行氧化性Heck交叉偶联的第一种通用方法,该方法还可以与其他杂环一起选择。


























 京公网安备 11010802027423号
京公网安备 11010802027423号