当前位置:
X-MOL 学术
›
Sustain. Energy Fuels
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
On the role of local heating in cathode degradation during the oxygen reduction reaction in solid acid fuel cells
Sustainable Energy & Fuels ( IF 5.6 ) Pub Date : 2020-08-21 , DOI: 10.1039/d0se00842g Maximilian Wagner 1, 2, 3, 4 , Oliver Lorenz 1, 2, 3, 4 , Felix P. Lohmann-Richters 4, 5, 6 , Aron Varga 4, 7, 8, 9 , Bernd Abel 1, 2, 3, 4
Sustainable Energy & Fuels ( IF 5.6 ) Pub Date : 2020-08-21 , DOI: 10.1039/d0se00842g Maximilian Wagner 1, 2, 3, 4 , Oliver Lorenz 1, 2, 3, 4 , Felix P. Lohmann-Richters 4, 5, 6 , Aron Varga 4, 7, 8, 9 , Bernd Abel 1, 2, 3, 4
Affiliation
|
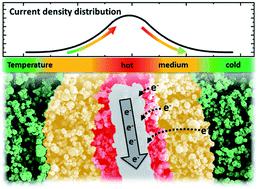
Reliable, stable, and long-term performance is one of the most important requirements for fuel cells in general. Widespread application of intermediate temperature solid acid fuel cells is still hindered by relatively fast degradation. However, durability studies are both expensive as well as, by their nature, time consuming and therefore rarely performed. In this study, we propose a viable method to investigate degradation pathways on a practical time scale. Five different types of electrodes were fabricated with varying geometrical complexity, but all containing platinum as the electrocatalyst. By utilizing small amounts of well-connected platinum as electrode catalyst, outstanding mass normalized currents were achieved resulting in accelerated cell degradation. Clearly observable effects on the electrodes were characterized ex situ by scanning electron microscopy and the electrochemical activity measured in operando by the decline of the current density at a constant cell voltage. After electrochemical measurement, changes of the electrodes were almost exclusively limited to the cathode side, where the electrolyte CsH2PO4 penetrated the previously distinct platinum layer originating from the current collector fibers. The observed morphological changes decreased the number of electrocatalytically active sites by covering the platinum layer or isolating the current collectors. These effects correlate both with the duration of the measurement and the current density. At different potentials, an asymptotic behavior of the cell performance was observed, identifying current-induced localized heating as the main degradation mechanism. Due to the high overpotential at the cathode, hotspots close to the current collectors could reach sufficient temperatures during cell operation to facilitate a morphological change of the electrolyte. This work gives a detailed analysis of the degradation mechanism in platinum-based solid acid fuel cell electrodes, providing valuable information for designing stable high-performance electrodes.
中文翻译:

固体酸燃料电池中氧还原反应过程中局部加热在阴极降解中的作用
总体上,可靠,稳定和长期的性能是燃料电池最重要的要求之一。相对快速的降解仍然阻碍了中温固体酸燃料电池的广泛应用。然而,耐久性研究既昂贵又因其性质而耗时,因此很少进行。在这项研究中,我们提出了一种可行的方法来研究在实际时间尺度上的降解途径。制造了五种不同类型的电极,它们的几何复杂度各不相同,但均包含铂作为电催化剂。通过使用少量连接良好的铂作为电极催化剂,可获得出色的质量归一化电流,从而加速了电池的降解。明确观察到对电极的影响易地通过扫描电子显微镜和测得的电化学活性在operando由电流密度的以恒定的电池电压的下降。在电化学测量之后,电极的变化几乎完全限于阴极侧,在此处电解质CsH 2 PO 4穿透了原先来自集电器纤维的独特铂层。通过覆盖铂层或隔离集电器,观察到的形态变化减少了电催化活性位点的数量。这些影响与测量持续时间和电流密度相关。在不同的电位下,观察到电池性能的渐近行为,确定电流诱导的局部加热是主要的降解机理。由于阴极处的高过电势,靠近集电器的热点在电池运行期间可能达到足够的温度,以促进电解质的形态变化。这项工作详细分析了铂基固体酸燃料电池电极的降解机理,
更新日期:2020-09-29
中文翻译:

固体酸燃料电池中氧还原反应过程中局部加热在阴极降解中的作用
总体上,可靠,稳定和长期的性能是燃料电池最重要的要求之一。相对快速的降解仍然阻碍了中温固体酸燃料电池的广泛应用。然而,耐久性研究既昂贵又因其性质而耗时,因此很少进行。在这项研究中,我们提出了一种可行的方法来研究在实际时间尺度上的降解途径。制造了五种不同类型的电极,它们的几何复杂度各不相同,但均包含铂作为电催化剂。通过使用少量连接良好的铂作为电极催化剂,可获得出色的质量归一化电流,从而加速了电池的降解。明确观察到对电极的影响易地通过扫描电子显微镜和测得的电化学活性在operando由电流密度的以恒定的电池电压的下降。在电化学测量之后,电极的变化几乎完全限于阴极侧,在此处电解质CsH 2 PO 4穿透了原先来自集电器纤维的独特铂层。通过覆盖铂层或隔离集电器,观察到的形态变化减少了电催化活性位点的数量。这些影响与测量持续时间和电流密度相关。在不同的电位下,观察到电池性能的渐近行为,确定电流诱导的局部加热是主要的降解机理。由于阴极处的高过电势,靠近集电器的热点在电池运行期间可能达到足够的温度,以促进电解质的形态变化。这项工作详细分析了铂基固体酸燃料电池电极的降解机理,

























 京公网安备 11010802027423号
京公网安备 11010802027423号