当前位置:
X-MOL 学术
›
Org. Biomol. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Synthesis of fused-tetrahydropyrimidines: one-pot methylenation-cyclization utilizing two molecules of CO2.
Organic & Biomolecular Chemistry ( IF 3.2 ) Pub Date : 2020-08-18 , DOI: 10.1039/d0ob01504k Yulei Zhao 1 , Xuqiang Guo , Yulan Du , Xinrui Shi , Shina Yan , Yunlin Liu , Jinmao You
Organic & Biomolecular Chemistry ( IF 3.2 ) Pub Date : 2020-08-18 , DOI: 10.1039/d0ob01504k Yulei Zhao 1 , Xuqiang Guo , Yulan Du , Xinrui Shi , Shina Yan , Yunlin Liu , Jinmao You
Affiliation
|
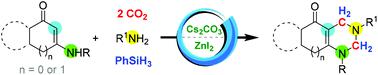
A methylenation–cyclization reaction, employing cyclic enaminones with primary aromatic amines and two molecules of CO2, furnishing fused-tetrahydropyrimidines, is discussed. In this Cs2CO3 and ZnI2 catalyzed one-pot two-step procedure, two molecules of CO2 were selectively converted to methylene groups. The multi-component reaction might proceed through the formation of bis(silyl)acetal which was followed by condensation and further aza-Diels–Alder reaction. Hydroquinazoline, hydrocyclopenta[d]pyrimidine and hydroindeno[1,2-d]pyrimidine derivatives could be prepared with CO2 as the C1 source, effectively.
中文翻译:

稠合四氢嘧啶的合成:利用两分子 CO2 的一锅亚甲基化环化。
讨论了使用环状烯胺酮与伯芳香胺和两个 CO 2分子,提供稠合四氢嘧啶的亚甲基化-环化反应。在这个Cs 2 CO 3和ZnI 2催化的一锅两步法中,两个CO 2分子被选择性地转化为亚甲基。多组分反应可能通过形成双(甲硅烷基)缩醛进行,然后是缩合和进一步的氮杂-狄尔斯-阿尔德反应。氢喹唑啉、氢环戊二烯[ d ]嘧啶和氢茚并[1,2- d ]嘧啶衍生物可以以CO 2作为C1源有效地制备。
更新日期:2020-09-16
中文翻译:

稠合四氢嘧啶的合成:利用两分子 CO2 的一锅亚甲基化环化。
讨论了使用环状烯胺酮与伯芳香胺和两个 CO 2分子,提供稠合四氢嘧啶的亚甲基化-环化反应。在这个Cs 2 CO 3和ZnI 2催化的一锅两步法中,两个CO 2分子被选择性地转化为亚甲基。多组分反应可能通过形成双(甲硅烷基)缩醛进行,然后是缩合和进一步的氮杂-狄尔斯-阿尔德反应。氢喹唑啉、氢环戊二烯[ d ]嘧啶和氢茚并[1,2- d ]嘧啶衍生物可以以CO 2作为C1源有效地制备。

























 京公网安备 11010802027423号
京公网安备 11010802027423号