Journal of Organometallic Chemistry ( IF 2.3 ) Pub Date : 2020-08-17 , DOI: 10.1016/j.jorganchem.2020.121482 Eduardo Laga , Carlos Cativiela , Esteban P. Urriolabeitia
|
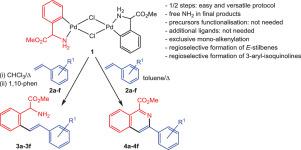
The ortho-palladated phenylglycine derivative [Pd(µ-Cl)(C6H4C(H)(CO2Me)NH2-C,N)]2 (1) reacts with substituted styrenes R1C6H4CH=CH2 (2a–2f) to give two different compounds as a function of the reaction conditions. When the reaction was performed in chloroform under reflux, the stilbene derivatives [C6H4-1-(CH(CO2Me)(NH2))-2-(E)-(CH=CHC6H4R1)] (3a–3f) were obtained, but when the reaction was carried out in toluene under reflux, the 3-arylisoquinoline-1-carboxylates (4a–4f) were obtained instead. The method has distinct advantages over other methods in that it is simple and general, only one/two reaction steps are necessary to obtain the final product, the prefunctionalisation of the substrates is not mandatory and nor is the presence of ancillary ligands, and a regioselective mono-insertion of the alkenes is systematically observed.
中文翻译:

邻palpalated苯基甘氨酸的烯基化:二苯乙烯衍生物和3-芳基-异喹啉-1-羧酸酯的合成
的邻位-palladated苯基甘氨酸衍生物[钯(μ-Cl)的(C 6 H ^ 4 C(H)(CO 2 Me)的NH 2 -C,N)] 2(1)反应以取代苯乙烯- [R 1 C ^ 6 ħ 4 CH = CH 2(2a - 2f)根据反应条件给出两种不同的化合物。当反应在氯仿中回流下进行时,二苯乙烯衍生物[C 6 H 4 -1-(CH(CO 2 Me)(NH 2))-2-(E)-(CH = CHC 6 H 4获得了R 1)](3a – 3f),但是当反应在甲苯中在回流条件下进行时,取而代之的是获得了3-芳基异喹啉-1-羧酸盐(4a - 4f)。该方法相对于其他方法具有明显的优势,因为它简单且通用,仅需一个/两个反应步骤即可获得最终产物,底物的预功能化不是强制性的,辅助配体的存在也不是必需的,并且具有区域选择性系统地观察到烯烃的单插入。



























 京公网安备 11010802027423号
京公网安备 11010802027423号