Journal of Allergy and Clinical Immunology ( IF 14.2 ) Pub Date : 2020-08-11 , DOI: 10.1016/j.jaci.2020.08.001 Anabel Gonzalez-Gil 1 , T August Li 1 , Ryan N Porell 1 , Steve M Fernandes 1 , Haley E Tarbox 1 , Hyun Sil Lee 2 , Kazuhiro Aoki 3 , Michael Tiemeyer 3 , Jean Kim 4 , Ronald L Schnaar 1
|
Background
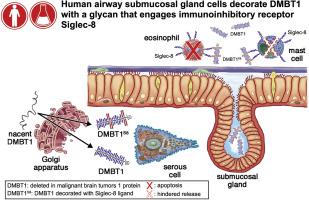
The immunoinhibitory receptor Siglec-8 on the surface of human eosinophils and mast cells binds to sialic acid–containing ligands in the local milieu, resulting in eosinophil apoptosis, inhibition of mast cell degranulation, and suppression of inflammation. Siglec-8 ligands were found on postmortem human trachea and bronchi and on upper airways in 2 compartments, cartilage and submucosal glands, but they were surprisingly absent from the epithelium. We hypothesized that Siglec-8 ligands in submucosal glands and ducts are normally transported to the airway mucus layer, which is lost during tissue preparation.
Objective
Our aim was to identify the major Siglec-8 sialoglycan ligand on the mucus layer of human airways.
Methods
Human upper airway mucus layer proteins were recovered during presurgical nasal lavage of patients at a sinus clinic. Proteins were resolved by gel electrophoresis and blotted, and Siglec-8 ligands detected. Ligands were purified by size exclusion and affinity chromatography, identified by proteomic mass spectrometry, and validated by electrophoretic and histochemical colocalization. The affinity of Siglec-8 binding to purified human airway ligand was determined by inhibition of glycan binding.
Results
A Siglec-8-ligand with a molecular weight of approximately 1000 kDa was found in all patient nasal lavage samples. Purification and identification revealed deleted in malignant brain tumors 1 (DMBT1) (also known by the aliases GP340 and SALSA), a large glycoprotein with multiple O-glycosylation repeats. Immunoblotting, immunohistochemistry, and enzyme treatments confirmed that Siglec-8 ligand on the human airway mucus layer is an isoform of DMBT1 carrying O-linked sialylated keratan sulfate chains (DMBT1S8). Quantitative inhibition revealed that DMBT1S8 has picomolar affinity for Siglec-8.
Conclusion
A distinct DMBT1 isoform, DMBT1S8, is the major high-avidity ligand for Siglec-8 on human airways.
中文翻译:

嗜酸性粒细胞和肥大细胞免疫抑制受体 Siglec-8 的人气道配体的分离、鉴定和表征
背景
人嗜酸性粒细胞和肥大细胞表面的免疫抑制受体 Siglec-8 与局部环境中含唾液酸的配体结合,导致嗜酸性粒细胞凋亡、抑制肥大细胞脱粒和抑制炎症。Siglec-8 配体在死后的人气管和支气管以及 2 个隔室、软骨和粘膜下腺的上呼吸道中发现,但令人惊讶的是,它们在上皮中不存在。我们假设黏膜下腺和导管中的 Siglec-8 配体通常被转运到气道黏液层,在组织制备过程中会丢失。
客观的
我们的目的是鉴定人类气道粘液层上的主要 Siglec-8 唾液酸聚糖配体。
方法
在鼻窦诊所对患者进行术前洗鼻期间回收了人上气道粘液层蛋白。通过凝胶电泳和印迹分离蛋白质,并检测到 Siglec-8 配体。通过尺寸排阻和亲和色谱纯化配体,通过蛋白质组质谱鉴定,并通过电泳和组织化学共定位进行验证。Siglec-8 与纯化的人气道配体结合的亲和力是通过抑制聚糖结合来确定的。
结果
在所有患者鼻腔灌洗液样品中均发现了分子量约为 1000 kDa 的 Siglec-8 配体。纯化和鉴定发现在恶性脑肿瘤 1 (DMBT1)(也称为别名 GP340 和 SALSA)中缺失,这是一种具有多个 O-糖基化重复序列的大型糖蛋白。免疫印迹、免疫组织化学和酶处理证实,人气道粘液层上的 Siglec-8 配体是 DMBT1 的同种型,携带 O-连接的唾液酸化硫酸角质素链 (DMBT1 S8 )。定量抑制显示 DMBT1 S8对 Siglec-8 具有皮摩尔亲和力。
结论
一种独特的 DMBT1 异构体 DMBT1 S8是 Siglec-8 在人体气道上的主要高亲和力配体。

























 京公网安备 11010802027423号
京公网安备 11010802027423号