当前位置:
X-MOL 学术
›
Acta Cryst. F
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Structural analysis of a novel substrate-free form of the aminoglycoside 6'-N-acetyltransferase from Enterococcus faecium.
Acta Crystallographica Section F ( IF 1.072 ) Pub Date : 2020-08-03 , DOI: 10.1107/s2053230x20009735 Hyunseok Jang 1 , Sunghark Kwon 1 , Chang Sook Jeong 2 , Chang Woo Lee 2 , Jisub Hwang 2 , Kyoung Ho Jung 1 , Jun Hyuck Lee 2 , Hyun Ho Park 1
Acta Crystallographica Section F ( IF 1.072 ) Pub Date : 2020-08-03 , DOI: 10.1107/s2053230x20009735 Hyunseok Jang 1 , Sunghark Kwon 1 , Chang Sook Jeong 2 , Chang Woo Lee 2 , Jisub Hwang 2 , Kyoung Ho Jung 1 , Jun Hyuck Lee 2 , Hyun Ho Park 1
Affiliation
|
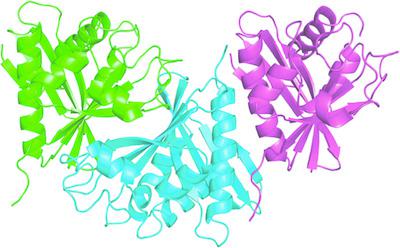
Aminoglycoside acetyltransferases (AACs) catalyze the transfer of an acetyl group between acetyl‐CoA and an aminoglycoside, producing CoA and an acetylated aminoglycoside. AAC(6′)‐Ii enzymes target the amino group linked to the 6′ C atom in an aminoglycoside. Several structures of the AAC(6′)‐Ii from Enterococcus faecium [Ef‐AAC(6′)‐Ii] have been reported to date. However, the detailed mechanism of its enzymatic function remains elusive. In this study, the crystal structure of Ef‐AAC(6′)‐Ii was determined in a novel substrate‐free form. Based on structural analysis, it is proposed that Ef‐AAC(6′)‐Ii sequentially undergoes conformational selection and induced fit for substrate binding. These results therefore provide a novel viewpoint on the mechanism of action of Ef‐AAC(6′)‐Ii.
中文翻译:

来自屎肠球菌的氨基糖苷 6'-N-乙酰转移酶的新型无底物形式的结构分析。
氨基糖苷乙酰转移酶 (AAC) 催化乙酰辅酶 A 和氨基糖苷之间的乙酰基转移,产生 CoA 和乙酰化氨基糖苷。AAC(6')-Ii 酶靶向与氨基糖苷中 6'C 原子相连的氨基。迄今为止,已报道了屎肠球菌[Ef-AAC(6')-Ii]的 AAC(6')-Ii 的几种结构。然而,其酶功能的详细机制仍然难以捉摸。在这项研究中,以一种新型的无底物形式确定了 Ef-AAC(6')-Ii 的晶体结构。基于结构分析,提出 Ef-AAC(6')-Ii 依次经历构象选择和诱导适合底物结合。因此,这些结果为 Ef-AAC(6')-Ii 的作用机制提供了新的观点。
更新日期:2020-08-03
中文翻译:

来自屎肠球菌的氨基糖苷 6'-N-乙酰转移酶的新型无底物形式的结构分析。
氨基糖苷乙酰转移酶 (AAC) 催化乙酰辅酶 A 和氨基糖苷之间的乙酰基转移,产生 CoA 和乙酰化氨基糖苷。AAC(6')-Ii 酶靶向与氨基糖苷中 6'C 原子相连的氨基。迄今为止,已报道了屎肠球菌[Ef-AAC(6')-Ii]的 AAC(6')-Ii 的几种结构。然而,其酶功能的详细机制仍然难以捉摸。在这项研究中,以一种新型的无底物形式确定了 Ef-AAC(6')-Ii 的晶体结构。基于结构分析,提出 Ef-AAC(6')-Ii 依次经历构象选择和诱导适合底物结合。因此,这些结果为 Ef-AAC(6')-Ii 的作用机制提供了新的观点。

























 京公网安备 11010802027423号
京公网安备 11010802027423号