当前位置:
X-MOL 学术
›
Adv. Synth. Catal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
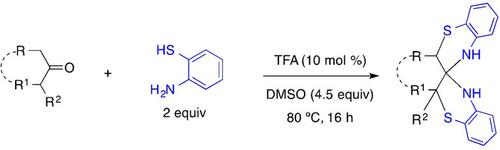
Umpolung Strategy for α,α’‐Functionalization of Ketones with 2‐Aminothiophenols: Stereoselective Access to Spirobis(1,4‐benzothiazines)
Advanced Synthesis & Catalysis ( IF 5.4 ) Pub Date : 2020-07-16 , DOI: 10.1002/adsc.202000725 Thanh Binh Nguyen 1 , Pascal Retailleau 1
Advanced Synthesis & Catalysis ( IF 5.4 ) Pub Date : 2020-07-16 , DOI: 10.1002/adsc.202000725 Thanh Binh Nguyen 1 , Pascal Retailleau 1
Affiliation
|
In the presence of TFA as a strong acid catalyst in DMSO, α,α’‐enolizable ketones were found to be stereoselectively α,α’‐functionalized with 2‐aminothiophenols to provide spirobis(1,4‐benzothiazine) derivatives.
中文翻译:

具有2-氨基硫酚的酮的α,α'-官能化的Umpolung策略:立体选择性获得Spirobis(1,4-苯并噻嗪)
在TFA作为DMSO中的强酸催化剂存在下,发现α,α'可烯化的酮被2-氨基苯硫酚立体选择性地进行α,α'-官能化,以提供螺双(1,4-苯并噻嗪)衍生物。
更新日期:2020-09-21
中文翻译:

具有2-氨基硫酚的酮的α,α'-官能化的Umpolung策略:立体选择性获得Spirobis(1,4-苯并噻嗪)
在TFA作为DMSO中的强酸催化剂存在下,发现α,α'可烯化的酮被2-氨基苯硫酚立体选择性地进行α,α'-官能化,以提供螺双(1,4-苯并噻嗪)衍生物。



























 京公网安备 11010802027423号
京公网安备 11010802027423号