当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
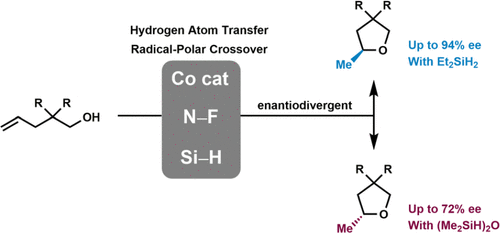
Catalyst- and Silane-Controlled Enantioselective Hydrofunctionalization of Alkenes by Cobalt-Catalyzed Hydrogen Atom Transfer and Radical-Polar Crossover
Journal of the American Chemical Society ( IF 15.0 ) Pub Date : 2020-07-10 , DOI: 10.1021/jacs.0c05017 Kousuke Ebisawa 1 , Kana Izumi 1 , Yuka Ooka 1 , Hiroaki Kato 1 , Sayori Kanazawa 1 , Sayura Komatsu 1 , Eriko Nishi 1 , Hiroki Shigehisa 1
Journal of the American Chemical Society ( IF 15.0 ) Pub Date : 2020-07-10 , DOI: 10.1021/jacs.0c05017 Kousuke Ebisawa 1 , Kana Izumi 1 , Yuka Ooka 1 , Hiroaki Kato 1 , Sayori Kanazawa 1 , Sayura Komatsu 1 , Eriko Nishi 1 , Hiroki Shigehisa 1
Affiliation
|
Catalytic enantioselective synthesis of tetrahydrofurans, which are found in the structures of many biologically active natural products, via a transition-metal catalyzed-hydrogen atom transfer (TM-HAT) and radical-polar crossover (RPC) mechanism is described herein. Hydroalkoxylation of non-conjugated alkenes proceeded efficiently with excellent enantioselectivity (up to 94% ee) using a suitable chiral cobalt catalyst, N-fluoro-2,4,6-collidinium tetrafluoroborate, and diethylsilane. Surprisingly, absolute configuration of the product was highly dependent on the steric hindrance of the silane. Slow addition of the silane, the dioxygen effect in the solvent, thermal dependency, and DFT calculation results supported the unprecedented scenario of two competing selective mechanisms. For the less-hindered diethylsilane, a high concentration of diffused carbon-centered radicals invoked diastereoenrichment of an alkylcobalt(III) intermediate by a radical chain reaction, which eventually determined the absolute configuration of the product. On the other hand, a more hindered silane resulted in less opportunity for radical chain reaction, instead facilitating enantioselective kinetic resolution during the late-stage nucleophilic displacement of the alkylcobalt(IV) intermediate.
中文翻译:

通过钴催化氢原子转移和自由基极性交叉对烯烃进行催化剂和硅烷控制的对映选择性加氢官能化
本文描述了通过过渡金属催化氢原子转移 (TM-HAT) 和自由基极性交叉 (RPC) 机制催化对映选择性合成四氢呋喃,四氢呋喃存在于许多生物活性天然产物的结构中。使用合适的手性钴催化剂、N-fluoro-2,4,6-collidinium 四氟硼酸盐和二乙基硅烷,非共轭烯烃的氢烷氧基化以优异的对映选择性(高达 94% ee)高效进行。令人惊讶的是,产品的绝对构型高度依赖于硅烷的空间位阻。硅烷的缓慢添加、溶剂中的双氧效应、热相关性和 DFT 计算结果支持了两种竞争选择机制的前所未有的情况。对于受阻较少的二乙基硅烷,高浓度扩散的以碳为中心的自由基通过自由基链反应引起烷基钴 (III) 中间体的非对映富集,最终决定了产品的绝对构型。另一方面,在烷基钴 (IV) 中间体的后期亲核置换过程中,受阻程度更高的硅烷导致自由基链反应的机会更少,反而促进了对映选择性动力学拆分。
更新日期:2020-07-10
中文翻译:

通过钴催化氢原子转移和自由基极性交叉对烯烃进行催化剂和硅烷控制的对映选择性加氢官能化
本文描述了通过过渡金属催化氢原子转移 (TM-HAT) 和自由基极性交叉 (RPC) 机制催化对映选择性合成四氢呋喃,四氢呋喃存在于许多生物活性天然产物的结构中。使用合适的手性钴催化剂、N-fluoro-2,4,6-collidinium 四氟硼酸盐和二乙基硅烷,非共轭烯烃的氢烷氧基化以优异的对映选择性(高达 94% ee)高效进行。令人惊讶的是,产品的绝对构型高度依赖于硅烷的空间位阻。硅烷的缓慢添加、溶剂中的双氧效应、热相关性和 DFT 计算结果支持了两种竞争选择机制的前所未有的情况。对于受阻较少的二乙基硅烷,高浓度扩散的以碳为中心的自由基通过自由基链反应引起烷基钴 (III) 中间体的非对映富集,最终决定了产品的绝对构型。另一方面,在烷基钴 (IV) 中间体的后期亲核置换过程中,受阻程度更高的硅烷导致自由基链反应的机会更少,反而促进了对映选择性动力学拆分。


























 京公网安备 11010802027423号
京公网安备 11010802027423号