当前位置:
X-MOL 学术
›
Angew. Chem. Int. Ed.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
A General Approach to O-Sulfation by a Sulfur(VI) Fluoride Exchange Reaction.
Angewandte Chemie International Edition ( IF 16.6 ) Pub Date : 2020-07-09 , DOI: 10.1002/anie.202007211 Chao Liu 1 , Cangjie Yang 1 , Seung Hwang 1 , Samantha L Ferraro 1 , James P Flynn 1 , Jia Niu 1
Angewandte Chemie International Edition ( IF 16.6 ) Pub Date : 2020-07-09 , DOI: 10.1002/anie.202007211 Chao Liu 1 , Cangjie Yang 1 , Seung Hwang 1 , Samantha L Ferraro 1 , James P Flynn 1 , Jia Niu 1
Affiliation
|
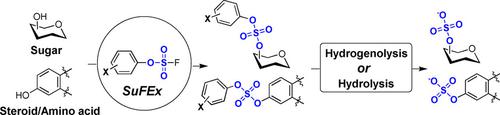
O‐sulfation is an important chemical code widely existing in bioactive molecules, but the scalable and facile synthesis of complex bioactive molecules carrying O‐sulfates remains challenging. Reported here is a general approach to O‐sulfation by the sulfur(VI) fluoride exchange (SuFEx) reaction between aryl fluorosulfates and silylated hydroxy groups. Efficient sulfate diester formation was achieved through systematic optimization of the electronic properties of aryl fluorosulfates. The versatility of this O‐sulfation strategy was demonstrated in the scalable syntheses of a variety of complex molecules carrying sulfate diesters at various positions, including monosaccharides, disaccharides, an amino acid, and a steroid. Selective hydrolytic and hydrogenolytic removal of the aryl masking groups from sulfate diesters yielded the corresponding O‐sulfate products in excellent yields. This strategy provides a powerful tool for the synthesis of O‐sulfate bioactive compounds.
中文翻译:

通过硫 (VI) 氟化物交换反应进行 O-硫酸化的一般方法。
O-硫酸化是生物活性分子中广泛存在的重要化学代码,但携带 O-硫酸盐的复杂生物活性分子的可扩展和简便合成仍然具有挑战性。这里报道的是通过芳基氟硫酸盐和甲硅烷基化羟基之间的硫 (VI) 氟化物交换 (SuFEx) 反应进行 O-硫酸化的一般方法。通过对芳基氟硫酸盐的电子特性进行系统优化,实现了有效的硫酸二酯形成。这种 O-硫酸化策略的多功能性在各种复杂分子的可扩展合成中得到证明,这些分子在不同位置携带硫酸二酯,包括单糖、二糖、氨基酸和类固醇。从硫酸二酯中选择性水解和氢解去除芳基掩蔽基团,以优异的产率得到相应的 O-硫酸盐产物。该策略为合成 O-硫酸盐生物活性化合物提供了强大的工具。
更新日期:2020-07-09
中文翻译:

通过硫 (VI) 氟化物交换反应进行 O-硫酸化的一般方法。
O-硫酸化是生物活性分子中广泛存在的重要化学代码,但携带 O-硫酸盐的复杂生物活性分子的可扩展和简便合成仍然具有挑战性。这里报道的是通过芳基氟硫酸盐和甲硅烷基化羟基之间的硫 (VI) 氟化物交换 (SuFEx) 反应进行 O-硫酸化的一般方法。通过对芳基氟硫酸盐的电子特性进行系统优化,实现了有效的硫酸二酯形成。这种 O-硫酸化策略的多功能性在各种复杂分子的可扩展合成中得到证明,这些分子在不同位置携带硫酸二酯,包括单糖、二糖、氨基酸和类固醇。从硫酸二酯中选择性水解和氢解去除芳基掩蔽基团,以优异的产率得到相应的 O-硫酸盐产物。该策略为合成 O-硫酸盐生物活性化合物提供了强大的工具。


























 京公网安备 11010802027423号
京公网安备 11010802027423号