Sustainable Chemistry and Pharmacy ( IF 6 ) Pub Date : 2020-07-03 , DOI: 10.1016/j.scp.2020.100281 Maria Ricciardi , Raffaele Cucciniello , Joel Barrault , Antonio Faggiano , Carmine Capacchione , Antonio Proto
|
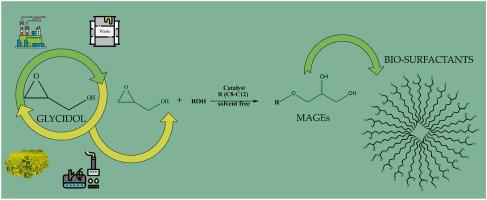
In this work we present a green synthetic pathway for the obtaining of bio-surfactants derived from glycerol. By extending a Lewis acid catalyzed reaction between glycidol and primary alcohols we obtained good conversion to the desired monoalkylglycidyl ether (MAGE) for long chain alcohols (n-octanol, n-decanol and n-dodecanol) reaching initial turnover frequency TOF up to 2633 h−1in the case of octanol. The obtained MAGEs were fully characterized and the critical micelle concentration (CMC) was determined for all bio-surfactants using a spectrophotometric approach such as pyrene method with peak difference (P2–P7) linear fitting (octylglyceryl ether: 0.97 ± 0.02 mM; decylglyceryl ether: 0.85 ± 0.03 mM; dodecylglyceryl ether: 0.71 ± 0.04 mM).
中文翻译:

迈向生物表面活性剂的一步:通过缩水甘油醇与Al(OTf)3催化的长链醇进行醇解合成单烷基甘油醚
在这项工作中,我们提出了一条绿色的合成途径,用于获得源自甘油的生物表面活性剂。通过扩展路易斯酸在缩水甘油和伯醇之间的催化反应,我们获得了长链醇(正辛醇,正癸醇和正十二烷醇)良好的转化率,使其转化为所需的单烷基缩水甘油醚(MAGE),初始转换频率TOF高达2633 h在辛醇的情况下为-1。对获得的MAGE进行了充分表征,并使用分光光度法(例如,method峰法(P2-P7)线性拟合(辛基甘油醚:0.97±0.02 mM;癸基甘油醚)等分光光度法测定所有生物表面活性剂的临界胶束浓度(CMC)。 :0.85±0.03mM;十二烷基甘油醚:0.71±0.04mM。

























 京公网安备 11010802027423号
京公网安备 11010802027423号