International Journal of Greenhouse Gas Control ( IF 3.9 ) Pub Date : 2020-07-02 , DOI: 10.1016/j.ijggc.2020.103102 Hanna Kierzkowska-Pawlak , Katarzyna Sobala
|
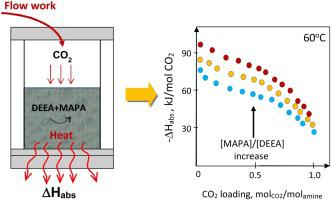
The heat of absorption is an important parameter that affects the CO2 absorption and stripping performance of amine-based CO2 capture. The present paper aims at development of the experimental method of determining the heat of absorption using a batch reaction calorimetry. A prospective high‐capacity CO2‐capturing solvent based on an aqueous solution of N,N-diethylethanolamine (DEEA) blended with small concentrations of 3-(methyloamino)propylamine (MAPA) is therefore studied. The differential and integral heat of absorption of CO2 was measured at 40 °C and 60 °C using single DEEA and amine blends. Experiments show that the heat of absorption in DEEA and DEEA + MAPA blends depends both on loading and temperature, though the temperature dependency is not as high as reported earlier. Increasing the MAPA/DEEA concentration ratio increased the heat of absorption in the amine blends.
中文翻译:

DEEA和DEEA + MAPA共混物水溶液中的CO 2吸收热-一种新的测量方法
吸收热是影响胺基CO 2捕集的CO 2吸收和汽提性能的重要参数。本文旨在开发一种使用间歇反应量热法测定吸收热的实验方法。因此,研究了一种基于N,N-二乙基乙醇胺(DEEA)与少量浓度的3-(甲氨基氨基)丙胺(MAPA)混合的高容量CO 2捕获溶剂。吸收CO 2的微分和积分热使用单一的DEEA和胺类混合物在40°C和60°C下测量。实验表明,DEEA和DEEA + MAPA共混物中的吸收热取决于载荷和温度,尽管温度依赖性不如先前报道的高。增加MAPA / DEEA的浓度比会增加胺共混物中的吸收热。


























 京公网安备 11010802027423号
京公网安备 11010802027423号