Electrochimica Acta ( IF 6.6 ) Pub Date : 2020-06-26 , DOI: 10.1016/j.electacta.2020.136601 Gilberto Rocha-Ortiz , Malcolm E. Tessensohn , Magali Salas-Reyes , Roberto Flores-Moreno , Richard D. Webster , Pablo D. Astudillo-Sánchez
|
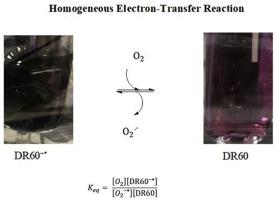
In this work, the voltammetric behaviour of disperse red 60 (DR60) in acetonitrile solutions in the presence of water and dioxygen (O2) is reported for the first time. DR60 can be reduced in two one-electron steps to form first the anion radical and then the dianion. The first reduction process of DR60 is affected by the water content of the acetonitrile, with digital simulations of the cyclic voltammetric waves supporting interactions between the radical anion and water molecules. Additionally, the presence of O2 modifies the first voltammetric process of DR60, suggesting the occurrence of homogeneous electron transfer reaction between anion radicals of DR60 and O2. Furthermore, the water content of the acetonitrile also affects the reduction wave of O2, with digital simulations supporting the occurrence of two association steps with the superoxide intermediate. The homogeneous electron transfer reaction between the quinone and oxygen species was also examined by reacting bulk electrolyzed solutions containing the anions of DR60 and O2 with neutral species of O2 and DR60, respectively, and recording the variation in the colour of the electrolyzed solutions. An equilibrium constant value was obtained from the difference between the formal reduction potentials of DR60 and O2 (−0.9955 and −0.9770, respectively, both vs. Ag in CH3CN with 0.5 M n-Bu4NPF6), indicating the direction of the homogenous electron transfer reaction (Keq = 2.013). Similarly, to confirm the occurrence of the homogeneous electron transfer reaction between O2 and another quinone based molecule, cyclic voltammetry and bulk electrolysis experiments were carried out on 9,10-anthraquinone (AQ), and its equilibrium constant value was also obtained (Keq = 4.392).
中文翻译:

乙腈溶液中蒽醌衍生物的阴离子种类与分子氧的均相电子转移反应:分散红60的电化学性质
在这项工作中,首次报道了分散红60(DR60)在乙腈溶液中在水和双氧(O 2)存在下的伏安行为。可以通过两个单电子步骤还原DR60,首先形成阴离子自由基,然后形成二价阴离子。DR60的第一个还原过程受乙腈的水含量影响,循环伏安波的数字模拟支持自由基阴离子和水分子之间的相互作用。另外,O 2的存在改变了DR60的第一个伏安过程,这表明DR60的阴离子自由基与O 2之间发生了均匀的电子转移反应。此外,乙腈的水含量也会影响O的还原波如图2所示,其中数字模拟支持与超氧化物中间体的两个缔合步骤的发生。还通过使含有DR60和O 2的阴离子的本体电解溶液分别与中性的O 2和DR60反应并记录电解质溶液的颜色变化来检查醌和氧之间的均匀电子转移反应。从DR60和O 2的形式还原电位之间的差异(分别为-0.9955和-0.9770,相对于具有0.5 M n -Bu 4 NPF 6的CH 3 CN中的Ag )获得平衡常数。)表示均匀电子转移反应的方向(K eq = 2.013)。同样,为确认O 2与另一种基于醌的分子之间发生均相电子转移反应,对9,10-蒽醌(AQ)进行了循环伏安法和本体电解实验,并获得了其平衡常数(K eq = 4.392)。

























 京公网安备 11010802027423号
京公网安备 11010802027423号