当前位置:
X-MOL 学术
›
Aging Cell
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Vitamin C is a source of oxoaldehyde and glycative stress in age-related cataract and neurodegenerative diseases.
Aging Cell ( IF 7.8 ) Pub Date : 2020-06-21 , DOI: 10.1111/acel.13176 Xingjun Fan 1, 2 , David R Sell 2 , Caili Hao 1 , Sabrina Liu 3 , Benlian Wang 4 , Daniel W Wesson 5 , Sandra Siedlak 2 , Xiongwei Zhu 2 , Terrance J Kavanagh 6 , Fiona E Harrison 7 , Vincent M Monnier 2, 8
Aging Cell ( IF 7.8 ) Pub Date : 2020-06-21 , DOI: 10.1111/acel.13176 Xingjun Fan 1, 2 , David R Sell 2 , Caili Hao 1 , Sabrina Liu 3 , Benlian Wang 4 , Daniel W Wesson 5 , Sandra Siedlak 2 , Xiongwei Zhu 2 , Terrance J Kavanagh 6 , Fiona E Harrison 7 , Vincent M Monnier 2, 8
Affiliation
|
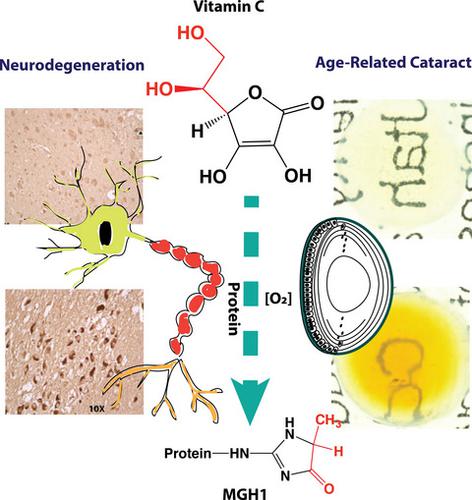
Oxoaldehyde stress has recently emerged as a major source of tissue damage in aging and age‐related diseases. The prevailing mechanism involves methylglyoxal production during glycolysis and modification of arginine residues through the formation of methylglyoxal hydroimidazolones (MG‐H1). We now tested the hypothesis that oxidation of vitamin C (ascorbic acid or ASA) contributes to this damage when the homeostatic redox balance is disrupted especially in ASA‐rich tissues such as the eye lens and brain. MG‐H1 measured by liquid chromatography mass spectrometry is several fold increased in the lens and brain from transgenic mice expressing human vitamin C transporter 2 (hSVCT2). Similarly, MG‐H1 levels are increased two‐ to fourfold in hippocampus extracts from individuals with Alzheimer's disease (AD), and significantly higher levels are present in sarkosyl‐insoluble tissue fractions from AD brain proteins than in the soluble fractions. Moreover, immunostaining with antibodies against methylglyoxal hydroimidazolones reveals similar increase in substantia nigra neurons from individuals with Parkinson's disease. Results from an in vitro incubation experiment suggest that accumulated catalytic metal ions in the hippocampus during aging could readily accelerate ASA oxidation and such acceleration was significantly enhanced in AD. Modeling studies and intraventricular injection of 13C‐labeled ASA revealed that ASA backbone carbons 4–6 are incorporated into MG‐H1 both in vitro and in vivo, likely via a glyceraldehyde precursor. We propose that drugs that prevent oxoaldehyde stress or excessive ASA oxidation may protect against age‐related cataract and neurodegenerative diseases.
中文翻译:

维生素 C 是与年龄相关的白内障和神经退行性疾病中氧化醛和糖化应激的来源。
氧醛应激最近已成为衰老和年龄相关疾病中组织损伤的主要来源。主要机制涉及糖酵解过程中甲基乙二醛的产生以及通过形成甲基乙二醛氢咪唑啉酮(MG-H1)对精氨酸残基进行修饰。我们现在测试了这样的假设:当稳态氧化还原平衡被破坏时,维生素 C(抗坏血酸或 ASA)的氧化会导致这种损害,尤其是在富含 ASA 的组织(例如眼晶状体和大脑)中。通过液相色谱质谱法测量,表达人维生素 C 转运蛋白 2 (hSVCT2) 的转基因小鼠的晶状体和大脑中的 MG-H1 增加了几倍。类似地,阿尔茨海默病 (AD) 患者海马提取物中的 MG-H1 水平增加了两到四倍,并且 AD 脑蛋白中肌氨酰不溶性组织部分中的 MG-H1 水平显着高于可溶性部分。此外,用抗甲基乙二醛氢咪唑酮抗体进行的免疫染色显示,帕金森病患者的黑质神经元也有类似的增加。体外培养实验的结果表明,衰老过程中海马中积累的催化金属离子很容易加速 ASA 氧化,并且这种加速在 AD 中显着增强。模型研究和心室内注射13 C 标记的 ASA 表明,ASA 主链碳 4-6 在体外和体内都可能通过甘油醛前体并入 MG-H1。我们提出,预防氧化醛应激或 ASA 过度氧化的药物可以预防与年龄相关的白内障和神经退行性疾病。
更新日期:2020-06-21
中文翻译:

维生素 C 是与年龄相关的白内障和神经退行性疾病中氧化醛和糖化应激的来源。
氧醛应激最近已成为衰老和年龄相关疾病中组织损伤的主要来源。主要机制涉及糖酵解过程中甲基乙二醛的产生以及通过形成甲基乙二醛氢咪唑啉酮(MG-H1)对精氨酸残基进行修饰。我们现在测试了这样的假设:当稳态氧化还原平衡被破坏时,维生素 C(抗坏血酸或 ASA)的氧化会导致这种损害,尤其是在富含 ASA 的组织(例如眼晶状体和大脑)中。通过液相色谱质谱法测量,表达人维生素 C 转运蛋白 2 (hSVCT2) 的转基因小鼠的晶状体和大脑中的 MG-H1 增加了几倍。类似地,阿尔茨海默病 (AD) 患者海马提取物中的 MG-H1 水平增加了两到四倍,并且 AD 脑蛋白中肌氨酰不溶性组织部分中的 MG-H1 水平显着高于可溶性部分。此外,用抗甲基乙二醛氢咪唑酮抗体进行的免疫染色显示,帕金森病患者的黑质神经元也有类似的增加。体外培养实验的结果表明,衰老过程中海马中积累的催化金属离子很容易加速 ASA 氧化,并且这种加速在 AD 中显着增强。模型研究和心室内注射13 C 标记的 ASA 表明,ASA 主链碳 4-6 在体外和体内都可能通过甘油醛前体并入 MG-H1。我们提出,预防氧化醛应激或 ASA 过度氧化的药物可以预防与年龄相关的白内障和神经退行性疾病。

























 京公网安备 11010802027423号
京公网安备 11010802027423号