Journal of Catalysis ( IF 7.3 ) Pub Date : 2020-06-20 , DOI: 10.1016/j.jcat.2020.06.013 Brandon L. Foley , Aditya Bhan
|
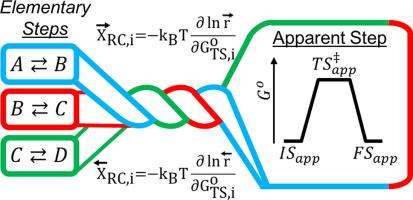
The net rate of a composite reaction is the difference between the forward and reverse reaction rates, which are kinetically distinct despite sharing elementary reaction steps and therefore have different rate-controlling transition states and species. Thus, degrees of rate control defined to identify rate-controlling transition states and species for the net rate confound contributions from the forward and reverse reactions. Herein, the forward and reverse degrees of rate control are defined to independently quantify the extent that species and transition states control the forward and reverse rates in reversible reactions. These degrees of rate control are defined as the relative change in the forward and reverse reaction rates per decrease in the standard-state molecular free energies of transition states and species, and they are related to experimentally measurable quantities such as steady-state fractional coverages, reaction orders, and energies and entropies of activation of the forward and reverse reaction rates. The forward/reverse degrees of rate control represent stoichiometric coefficients for species and transition states in an equilibrium between the initial states and transition states of the apparent rate-controlling steps of forward and reverse overall reactions. At equilibrium, the apparent transition states for the forward reaction and reverse reaction converge, and thus the forward and reverse rate-controlling steps combine to form a single apparent rate-controlling step. This apparent rate-controlling step is comprised of an apparent initial state, transition state, and final state, where the apparent final state of the forward reaction is the apparent initial state of the reverse reaction. The apparent rate-controlling step behaves identically to an elementary step reaction at equilibrium with a pseudo-mass-action rate function given by the transition-state-theory (TST) form rate function (Foley and Bhan, 2020) with a stoichiometric number equal to the affinity-averaged stoichiometric number, . The ratio of the forward and reverse TST-form rate functions is identical to the overall thermodynamic equilibrium relation, consistent with the principles of microscopic reversibility and detailed balance at equilibrium.
中文翻译:

可逆反应中热力学一致的正向和反向速率控制
复合反应的净速率是正向反应速率和反向反应速率之间的差异,尽管共享基本的反应步骤,但在动力学上是截然不同的,因此具有不同的速率控制过渡态和物种。因此,定义为识别速率控制的过渡状态和净速率的物种的速率控制程度混淆了正向和反向反应的贡献。在此,速率控制的正向和反向度被定义为独立地量化物质和过渡态在可逆反应中控制正向和反向率的程度。这些速率控制度定义为每反应的正向和反向反应速率的相对变化过渡态和物种的标准态分子自由能降低,并且与实验可测量的量有关,例如稳态分数覆盖率,反应阶数,正向和反向反应速率激活的能量和熵。速率控制的正向/反向度表示正向和反向总体反应的表观速率控制步骤的初始状态和过渡状态之间处于平衡状态时,物种和过渡状态的化学计量系数。在平衡时,正向反应和逆向反应的表观转变状态收敛,因此正向和反向速率控制步骤组合形成一个单一的表观速率控制步骤。这个表观速率控制步骤包括一个表观初始状态,过渡态和最终状态,其中正反应的表观最终状态是逆反应的表观初始状态。表观速率控制步骤在平衡时的行为与基本步骤反应相同,其化学假性速率函数由过渡态理论(TST)形式速率函数(Foley and Bhan,2020)给出,化学计量数等于到亲和力平均化学计量数,。TST形式速率函数的正向和反向比率与整体热力学平衡关系相同,这与微观可逆性原理和平衡时的详细平衡原理一致。

























 京公网安备 11010802027423号
京公网安备 11010802027423号