Bioorganic & Medicinal Chemistry Letters ( IF 2.7 ) Pub Date : 2020-06-10 , DOI: 10.1016/j.bmcl.2020.127341 Siva Hariprasad Kurma 1 , Shailaja Karri 2 , Madhusudana Kuncha 2 , Ramakrishna Sistla 2 , China Raju Bhimapaka 1
|
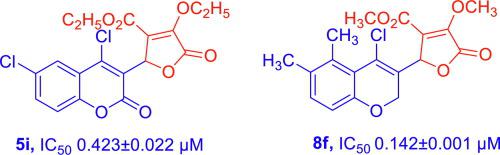
Cycloaddition reaction of 4-chloro-2-oxo-2H-chromene-3-carbaldehydes (3a-g) and 4-chloro-2H-chromene-3-carbaldehydes (7a-h) with activated alkynes (4a-b) provided the 2-oxo-2H-chromenyl-5-oxo-2,5-dihydrofuran-3-carboxylates (5a-n) and 2H-chromenyl-5-oxo-2,5-dihydrofuran-3-carboxylates (8a-p). All the prepared compounds were screened for anti-inflammatory activity. In vitro anti-inflammatory activity data demonstrated that the compounds 5g, 5i, 5k-l and 8f are effective among the tested compounds against TNF-α (1.108 ± 0.002, 0.423 ± 0.022, 0.047 ± 0.001, 0.070 ± 0.002 and 0.142 ± 0.001 µM) in comparison with standard compound Prednisolone (0.033 ± 0.002 µM). Based on in vitro results, three compounds (5i, 5k and 8f) have been selected for in vivo experiments and these compounds are identified as better compounds with respect to anti-inflammatory activity in LPS induced mice model. Compound 5i was identified as potent and showed significant reduction in TNF-α and IL-6.
中文翻译:

2-氧代-2H-苯并二苯并-2-H-色烯基-5-氧代-2,5-二氢呋喃-3-羧酸酯的合成及抗炎活性。
4-氯-2-氧代-2 H-亚甲基-3-甲醛(3a-g)和4-氯-2 H-亚甲基-3-甲醛(7a-h)与活化炔烃(4a-b)的环加成反应提供了2-oxo-2 H -chromenyl-5-oxo-2,5-dihydrofuran-3-羧酸盐(5a-n)和2 H -chromenyl-5-oxo-2,5-dihydrofuran-3-羧酸盐(8a -p)。筛选所有制备的化合物的抗炎活性。体外抗炎活性数据表明化合物5g,5i,5k-1和8f与标准化合物泼尼松龙(0.033±0.002 µM)相比,被测化合物对TNF-α(1.108±0.002,0.423±0.022,0.047±0.001,0.070±0.002和0.142±0.001 µM)有效。根据体外结果,已选择了三种化合物(5i,5k和8f)用于体内实验,在LPS诱导的小鼠模型中,就抗炎活性而言,这些化合物被确定为更好的化合物。化合物5i被确定为有效化合物,并显示出TNF-α和IL-6的显着降低。



























 京公网安备 11010802027423号
京公网安备 11010802027423号