Chemical Physics ( IF 2.3 ) Pub Date : 2020-06-04 , DOI: 10.1016/j.chemphys.2020.110873 Kiran Singh , Indu bala , Ramesh Kataria
|
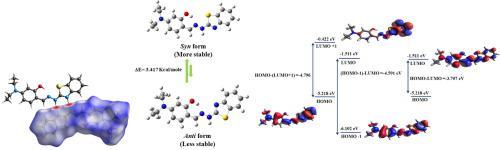
Benzothiazole based hydrazone was synthesised and characterised using FT-IR, 1H NMR & X-ray studies. The ground state geometry for the synthesised compound was predicted using density functional theory (DFT). The theoretically predicted structural parameters were found to be in close agreement with the experimentally determined. Intermolecular interactions present in the molecule were quantified using Hirshfeld surface analysis. Global reactivity descriptors were predicted to access the information related with the stability and reactivity of the molecule which shows the soft nature of the molecule. The sites available for electrophilic and nucleophilic attack were explored using local based descriptors in the form of condensed Fukui function. Hyperconjugative interactions present in the molecule were also quantified using Natural bond orbital (NBO) analysis of the molecule. Electrophilicity based charge transfer (ECT) study was done with DNA base pairs in order to explore the direction of charge transfer in the molecule
中文翻译:

苯并噻唑基的晶体结构,Hirshfeld表面和DFT基NBO,NLO,ECT和MEP
合成了基于苯并噻唑的并使用FT-IR,11 H NMR和X射线研究。使用密度泛函理论(DFT)预测了合成化合物的基态几何形状。发现理论上预测的结构参数与实验确定的参数非常一致。使用Hirshfeld表面分析对分子中存在的分子间相互作用进行定量。预测整体反应性描述符将访问与分子的稳定性和反应性有关的信息,该信息显示了分子的软性。亲和性和亲核性攻击的可用位点是使用基于局部的描述子以浓缩的Fukui函数形式进行探索的。还使用分子的自然键轨道(NBO)分析对分子中存在的超共轭相互作用进行了定量。



























 京公网安备 11010802027423号
京公网安备 11010802027423号