当前位置:
X-MOL 学术
›
J. Cell. Physiol.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Suppression of TRPM7 enhances TRAIL-induced apoptosis in triple-negative breast cancer cells.
Journal of Cellular Physiology ( IF 5.6 ) Pub Date : 2020-05-29 , DOI: 10.1002/jcp.29820 Chiman Song 1, 2 , Seunghye Choi 3 , Ki-Bong Oh 2 , Taebo Sim 1, 3, 4
Journal of Cellular Physiology ( IF 5.6 ) Pub Date : 2020-05-29 , DOI: 10.1002/jcp.29820 Chiman Song 1, 2 , Seunghye Choi 3 , Ki-Bong Oh 2 , Taebo Sim 1, 3, 4
Affiliation
|
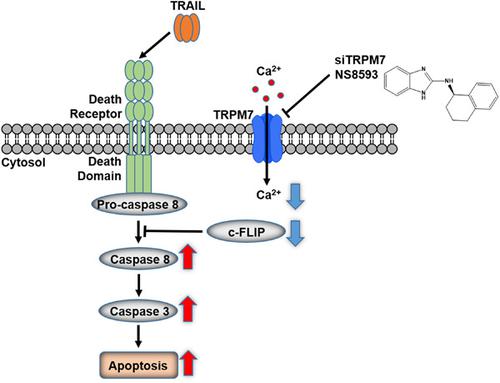
Transient receptor potential cation channel subfamily M member 7 (TRPM7) composed of an ion channel and a kinase domain regulates triple‐negative breast cancer (TNBC) cell migration, invasion, and metastasis, but it does not modulate TNBC proliferation. However, previous studies have shown that the combination treatment of nonselective TRPM7 channel inhibitors (2‐aminoethoxydiphenyl borate and Gd3+) with tumor necrosis factor‐related apoptosis‐inducing ligand (TRAIL) increases antiproliferative effects and apoptosis in prostate cancer cells and hepatic stellate cells. We, therefore, investigated the potential role of TRPM7 in proliferation and apoptosis of TNBC cells (MDA‐MB‐231 and MDA‐MB‐468 cells) with TRAIL. We demonstrated that suppression of TRPM7 via TRPM7 knockdown or pharmacological inhibition synergistically increases TRAIL‐induced antiproliferative effects and apoptosis in TNBC cells. Furthermore, we showed that the synergistic interaction might be associated with TRPM7 channel activities using combination treatments of TRAIL and TRPM7 inhibitors (NS8593 as a TRPM7 channel inhibitor and TG100‐115 as a TRPM7 kinase inhibitor). We reveal that downregulation of cellular FLICE‐inhibitory protein via inhibition of Ca2+ influx might be involved in the synergistic interaction. Our study would provide both a new role of TRPM7 in TNBC cell apoptosis and a potential combinatorial therapeutic strategy using TRPM7 inhibitors with TRAIL in the treatment of TNBC.
中文翻译:

TRPM7的抑制增强了三阴性乳腺癌细胞中TRAIL诱导的凋亡。
由离子通道和激酶结构域组成的瞬时受体潜在阳离子通道亚家族M成员7(TRPM7)调节三阴性乳腺癌(TNBC)细胞的迁移,侵袭和转移,但不调节TNBC增殖。然而,先前的研究表明,非选择性TRPM7通道抑制剂(2-氨基乙氧基二苯基硼酸盐和Gd 3+)与肿瘤坏死因子相关的凋亡诱导配体(TRAIL)的联合治疗可提高前列腺癌细胞和肝星状细胞的抗增殖作用和凋亡细胞。因此,我们研究了TRPM7在TRAIL对TNBC细胞(MDA-MB-231和MDA-MB-468细胞)增殖和凋亡中的潜在作用。我们证明了通过TRPM7抑制TRPM7抑制或药理抑制作用协同增加TNIL细胞中TRAIL诱导的抗增殖作用和细胞凋亡。此外,我们表明,使用TRAIL和TRPM7抑制剂(NS8593作为TRPM7通道抑制剂和TG100-115作为TRPM7激酶抑制剂)联合治疗,协同相互作用可能与TRPM7通道活性有关。我们揭示了通过抑制Ca 2+内流而使细胞FLICE抑制蛋白下调可能与协同相互作用有关。我们的研究将提供TRPM7在TNBC细胞凋亡中的新作用,以及将TRPM7抑制剂与TRAIL一起用于治疗TNBC的潜在组合治疗策略。
更新日期:2020-05-29
中文翻译:

TRPM7的抑制增强了三阴性乳腺癌细胞中TRAIL诱导的凋亡。
由离子通道和激酶结构域组成的瞬时受体潜在阳离子通道亚家族M成员7(TRPM7)调节三阴性乳腺癌(TNBC)细胞的迁移,侵袭和转移,但不调节TNBC增殖。然而,先前的研究表明,非选择性TRPM7通道抑制剂(2-氨基乙氧基二苯基硼酸盐和Gd 3+)与肿瘤坏死因子相关的凋亡诱导配体(TRAIL)的联合治疗可提高前列腺癌细胞和肝星状细胞的抗增殖作用和凋亡细胞。因此,我们研究了TRPM7在TRAIL对TNBC细胞(MDA-MB-231和MDA-MB-468细胞)增殖和凋亡中的潜在作用。我们证明了通过TRPM7抑制TRPM7抑制或药理抑制作用协同增加TNIL细胞中TRAIL诱导的抗增殖作用和细胞凋亡。此外,我们表明,使用TRAIL和TRPM7抑制剂(NS8593作为TRPM7通道抑制剂和TG100-115作为TRPM7激酶抑制剂)联合治疗,协同相互作用可能与TRPM7通道活性有关。我们揭示了通过抑制Ca 2+内流而使细胞FLICE抑制蛋白下调可能与协同相互作用有关。我们的研究将提供TRPM7在TNBC细胞凋亡中的新作用,以及将TRPM7抑制剂与TRAIL一起用于治疗TNBC的潜在组合治疗策略。

























 京公网安备 11010802027423号
京公网安备 11010802027423号