Tetrahedron ( IF 2.1 ) Pub Date : 2020-05-29 , DOI: 10.1016/j.tet.2020.131300 Toyoho Takeda , Airi Katayama , Yusuke Kinoshita , Hitoshi Tamiaki
|
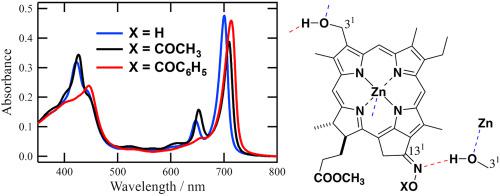
Zinc methyl 3-hydroxymethyl-pyropheophorbides-a possessing hydroxyimino, acetoxyimino, and benzoyloxyimino groups at the 131-position were prepared by chemical modification of naturally occurring chlorophyll-a. The (131E)- and (131Z)-stereoisomers of the obtained unsymmetric oximes were readily separated by column chromatography, and their stereochemistry was determined by 1H NMR spectroscopy. The synthetic O-(un)substituted oximes were monomeric in tetrahydrofuran and their electronic absorption spectra were dependent on the stereochemistry as well as the O-substituents in the oxime moiety. These chlorophyll-a derivatives self-aggregated in an aqueous Triton X-100 micellar solution to give red-shifted absorption bands. The J-aggregation ability of the (131E)-isomers was higher than that of the corresponding the (131Z)-isomers. Such stereoselective self-aggregation was comparable to that of bacteriochlorophylls-c, d, e, and f in the main light-harvesting antennas of green photosynthetic bacteria.
中文翻译:

锌13-肟功能化叶绿素a衍生物的合成及其(13 1 E / Z)依赖性自聚集
锌甲基-3-羟甲基pyropheophorbides-一个具有肟基,乙酰氧基亚氨基,和benzoyloxyimino在13组1是由天然存在的叶绿素化学改性制备的位上一个。所得不对称肟的(13 1 E)-和(13 1 Z)-立体异构体易于通过柱色谱法分离,并通过1 H NMR光谱法测定其立体化学。合成的O-(未)取代的肟在四氢呋喃中是单体,其电子吸收光谱取决于立体化学以及O肟部分中的-取代基。这些叶绿素一个衍生物自聚集在水的Triton X-100胶束溶液,得到红移吸收带。(13 1 E)异构体的J聚集能力高于相应的(13 1 Z)异构体的J聚集能力。这种立体选择性自聚集与绿色光合细菌的主要光收集天线中的细菌叶绿素-c,d,e和f相当。


























 京公网安备 11010802027423号
京公网安备 11010802027423号