当前位置:
X-MOL 学术
›
Angew. Chem. Int. Ed.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
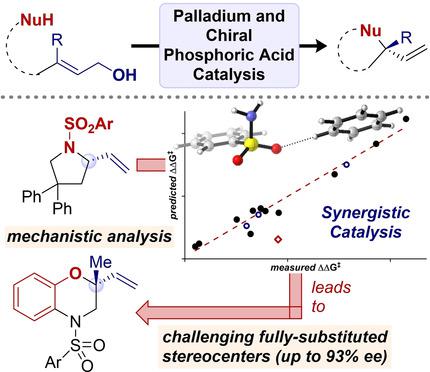
Enantioselective Intramolecular Allylic Substitution via Synergistic Palladium/Chiral Phosphoric Acid Catalysis: Insight into Stereoinduction through Statistical Modeling.
Angewandte Chemie International Edition ( IF 16.6 ) Pub Date : 2020-05-26 , DOI: 10.1002/anie.202006237 Cheng-Che Tsai,Christopher Sandford,Tao Wu,Buyun Chen,Matthew S Sigman,F Dean Toste
Angewandte Chemie International Edition ( IF 16.6 ) Pub Date : 2020-05-26 , DOI: 10.1002/anie.202006237 Cheng-Che Tsai,Christopher Sandford,Tao Wu,Buyun Chen,Matthew S Sigman,F Dean Toste
|
The mode of asymmetric induction in an enantioselective intramolecular allylic substitution reaction catalyzed by a combination of palladium and a chiral phosphoric acid was investigated by a combined experimental and statistical modeling approach. Experiments to probe nonlinear effects, the reactivity of deuterium‐labeled substrates, and control experiments revealed that nucleophilic attack to the π‐allylpalladium intermediate is the enantio‐determining step, in which the chiral phosphate anion is involved in stereoinduction. Using multivariable linear regression analysis, we determined that multiple noncovalent interactions with the chiral environment of the phosphate anion are integral to enantiocontrol in the transition state. The synthetic protocol to form chiral pyrrolidines was further applied to the asymmetric construction of C−O bonds at fully substituted carbon centers in the synthesis of chiral 2,2‐disubstituted benzomorpholines.
中文翻译:

通过协同钯/手性磷酸催化进行对映选择性分子内烯丙基取代:通过统计模型洞察立体诱导。
通过实验和统计建模相结合的方法研究了钯和手性磷酸组合催化的对映选择性分子内烯丙基取代反应中的不对称诱导模式。探测非线性效应、氘标记底物的反应性的实验和对照实验表明,对 π-烯丙基钯中间体的亲核攻击是对映体决定步骤,其中手性磷酸根阴离子参与立体诱导。使用多元线性回归分析,我们确定与磷酸根阴离子的手性环境的多种非共价相互作用对于过渡态的对映体控制是不可或缺的。形成手性吡咯烷的合成方案进一步应用于手性 2,2-二取代苯并吗啉合成中完全取代的碳中心处 C−O 键的不对称构建。
更新日期:2020-05-26
中文翻译:

通过协同钯/手性磷酸催化进行对映选择性分子内烯丙基取代:通过统计模型洞察立体诱导。
通过实验和统计建模相结合的方法研究了钯和手性磷酸组合催化的对映选择性分子内烯丙基取代反应中的不对称诱导模式。探测非线性效应、氘标记底物的反应性的实验和对照实验表明,对 π-烯丙基钯中间体的亲核攻击是对映体决定步骤,其中手性磷酸根阴离子参与立体诱导。使用多元线性回归分析,我们确定与磷酸根阴离子的手性环境的多种非共价相互作用对于过渡态的对映体控制是不可或缺的。形成手性吡咯烷的合成方案进一步应用于手性 2,2-二取代苯并吗啉合成中完全取代的碳中心处 C−O 键的不对称构建。

























 京公网安备 11010802027423号
京公网安备 11010802027423号