Current Protein & Peptide Science ( IF 2.8 ) Pub Date : 2020-08-31 , DOI: 10.2174/1389203721666200505101734 Mohd Saeed 1 , Mohd Adnan Kausar 2 , Rajeev Singh 3 , Arif J Siddiqui 1 , Asma Akhter 4
|
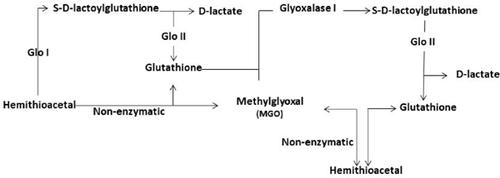
Glycation refers to the covalent binding of sugar molecules to macromolecules, such as DNA, proteins, and lipids in a non-enzymatic reaction, resulting in the formation of irreversibly bound products known as advanced glycation end products (AGEs). AGEs are synthesized in high amounts both in pathological conditions, such as diabetes and under physiological conditions resulting in aging. The body’s anti-glycation defense mechanisms play a critical role in removing glycated products. However, if this defense system fails, AGEs start accumulating, which results in pathological conditions. Studies have been shown that increased accumulation of AGEs acts as key mediators in multiple diseases, such as diabetes, obesity, arthritis, cancer, atherosclerosis, decreased skin elasticity, male erectile dysfunction, pulmonary fibrosis, aging, and Alzheimer’s disease. Furthermore, glycation of nucleotides, proteins, and phospholipids by α-oxoaldehyde metabolites, such as glyoxal (GO) and methylglyoxal (MGO), causes potential damage to the genome, proteome, and lipidome. Glyoxalase-1 (GLO-1) acts as a part of the anti-glycation defense system by carrying out detoxification of GO and MGO. It has been demonstrated that GLO-1 protects dicarbonyl modifications of the proteome and lipidome, thereby impeding the cell signaling and affecting age-related diseases. Its relationship with detoxification and anti-glycation defense is well established. Glycation of proteins by MGO and GO results in protein misfolding, thereby affecting their structure and function. These findings provide evidence for the rationale that the functional modulation of the GLO pathway could be used as a potential therapeutic target. In the present review, we summarized the newly emerged literature on the GLO pathway, including enzymes regulating the process. In addition, we described small bioactive molecules with the potential to modulate the GLO pathway, thereby providing a basis for the development of new treatment strategies against age-related complications.
中文翻译:

乙二醛酶在糖基化和羰基应激诱导的代谢紊乱中的作用。
糖基化是指糖分子在非酶反应中与DNA,蛋白质和脂质等大分子共价结合,导致形成不可逆结合的产物,称为高级糖化终产物(AGEs)。AGEs在诸如糖尿病的病理状况下以及在导致衰老的生理状况下都大量合成。人体的抗糖化防御机制在去除糖化产物中起着至关重要的作用。但是,如果此防御系统发生故障,AGEs将开始积累,从而导致病理状况。研究表明,AGEs积累的增加是多种疾病(例如糖尿病,肥胖,关节炎,癌症,动脉粥样硬化,皮肤弹性下降,男性勃起功能障碍,肺纤维化,衰老,和阿尔茨海默氏病。此外,诸如乙二醛(GO)和甲基乙二醛(MGO)之类的α-氧醛代谢产物对核苷酸,蛋白质和磷脂的糖基化作用可能对基因组,蛋白质组和脂质组造成潜在的损害。乙二醛酶1(GLO-1)通过对GO和MGO进行排毒而成为抗糖基化防御系统的一部分。已经证明GLO-1保护蛋白质组和脂质组的二羰基修饰,从而阻碍细胞信号传导并影响与年龄有关的疾病。它与排毒和抗糖基化防御的关系是众所周知的。MGO和GO的蛋白质糖基化导致蛋白质错误折叠,从而影响其结构和功能。这些发现为GLO途径的功能调节可用作潜在治疗靶点的理论依据提供了证据。在本综述中,我们总结了有关GLO途径的新兴文献,包括调节过程的酶。此外,我们描述了具有调节GLO途径潜力的小生物活性分子,从而为开发针对年龄相关并发症的新治疗策略提供了基础。


























 京公网安备 11010802027423号
京公网安备 11010802027423号