当前位置:
X-MOL 学术
›
React. Chem. Eng.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Nano-TiO2 promoted CaO-based high-temperature CO2 sorbent: influence of crystal level properties on the CO2 sorption efficiency
Reaction Chemistry & Engineering ( IF 3.9 ) Pub Date : 2020-05-05 , DOI: 10.1039/d0re00124d Sanat Chandra Maiti 1, 2, 3, 4, 5 , Chinmay Ghoroi 1, 2, 3, 4, 5
Reaction Chemistry & Engineering ( IF 3.9 ) Pub Date : 2020-05-05 , DOI: 10.1039/d0re00124d Sanat Chandra Maiti 1, 2, 3, 4, 5 , Chinmay Ghoroi 1, 2, 3, 4, 5
Affiliation
|
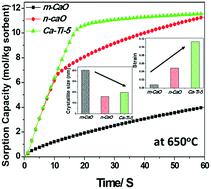
This work investigates the multi-cycle CO2 sorption and the kinetics of the carbonation reaction of nano-TiO2 promoted CaO synthesized from commercially available micron sized CaCO3. The morphology of CaCO3 coated with different wt% of nano-TiO2 (1 to 10 wt%) was investigated by field emission scanning electron microscopy (FESEM). The crystallite size, lattice parameters, and strain of the decomposed product for all the samples were estimated by Rietveld refinement of X-ray diffraction (XRD) data. The carbonation of nano-TiO2 promoted CaO sorbents was studied on a thermogravimetric analyzer (TG) under a CO2 atmosphere (0.02 MPa) at different temperatures (600, 650 and 700 °C) and the results were compared with the CaO obtained from pure micron sized CaCO3. The results show that the sorption capacity of nano-TiO2 promoted CaO sorbents is several times higher than that of the pure CaO. The improvement could be ascribed to the porous structure and smaller crystallite size of CaO in the presence of nano-TiO2, which is supported by FESEM and XRD. In fact, the sorption capacity of nano-TiO2 promoted CaO is found to be higher than that of the CaO obtained from nano-CaCO3. The shifting of the transition point between two-stage carbonation reactions is correlated with the wt% of nano-TiO2, the crystallite size, and the strain inside the CaO. The optimum wt% of nano-TiO2 and sorption temperature of the sorbents are explored to achieve the maximum sorption capacity. Finally, the experimental results are fitted with theoretical models based on the shrinking core model, and kinetic parameters are evaluated.
中文翻译:

纳米TiO2促进的CaO基高温CO2吸附剂:晶体水平性质对CO2吸附效率的影响
这项工作研究了多循环CO 2的吸附以及由市售微米级CaCO 3合成的纳米TiO 2促进的CaO碳酸化反应的动力学。通过场发射扫描电子显微镜(FESEM)研究了涂覆有不同重量%的纳米TiO 2(1至10重量%)的CaCO 3的形态。所有样品的微晶尺寸,晶格参数和分解产物的应变均通过Rietveld X射线衍射(XRD)数据精炼来估算。在热重分析仪(TG)上于CO 2下研究了纳米TiO 2促进的CaO吸附剂的碳酸化温度(600、650和700°C)在大气(0.02 MPa)下进行,并将结果与由纯微米尺寸的CaCO 3获得的CaO进行比较。结果表明,纳米TiO 2促进的CaO吸附剂的吸附能力是纯CaO的几倍。这种改善可以归因于在纳米TiO 2存在下CaO的多孔结构和较小的微晶尺寸,这是由FESEM和XRD支持的。实际上,发现纳米TiO 2促进的CaO的吸附能力高于从纳米CaCO 3获得的CaO的吸附能力。两阶段碳酸化反应之间的转变点的移动与纳米TiO 2的wt%相关,微晶尺寸和CaO内部的应变。探索了纳米TiO 2的最佳重量%和吸附剂的吸附温度,以实现最大吸附容量。最后,将实验结果与基于收缩核模型的理论模型进行拟合,并评估动力学参数。
更新日期:2020-06-30
中文翻译:

纳米TiO2促进的CaO基高温CO2吸附剂:晶体水平性质对CO2吸附效率的影响
这项工作研究了多循环CO 2的吸附以及由市售微米级CaCO 3合成的纳米TiO 2促进的CaO碳酸化反应的动力学。通过场发射扫描电子显微镜(FESEM)研究了涂覆有不同重量%的纳米TiO 2(1至10重量%)的CaCO 3的形态。所有样品的微晶尺寸,晶格参数和分解产物的应变均通过Rietveld X射线衍射(XRD)数据精炼来估算。在热重分析仪(TG)上于CO 2下研究了纳米TiO 2促进的CaO吸附剂的碳酸化温度(600、650和700°C)在大气(0.02 MPa)下进行,并将结果与由纯微米尺寸的CaCO 3获得的CaO进行比较。结果表明,纳米TiO 2促进的CaO吸附剂的吸附能力是纯CaO的几倍。这种改善可以归因于在纳米TiO 2存在下CaO的多孔结构和较小的微晶尺寸,这是由FESEM和XRD支持的。实际上,发现纳米TiO 2促进的CaO的吸附能力高于从纳米CaCO 3获得的CaO的吸附能力。两阶段碳酸化反应之间的转变点的移动与纳米TiO 2的wt%相关,微晶尺寸和CaO内部的应变。探索了纳米TiO 2的最佳重量%和吸附剂的吸附温度,以实现最大吸附容量。最后,将实验结果与基于收缩核模型的理论模型进行拟合,并评估动力学参数。

























 京公网安备 11010802027423号
京公网安备 11010802027423号