Chemical Physics ( IF 2.3 ) Pub Date : 2020-04-28 , DOI: 10.1016/j.chemphys.2020.110823 Yue Zhang , Zhongjie Han , Chunhua Li
|
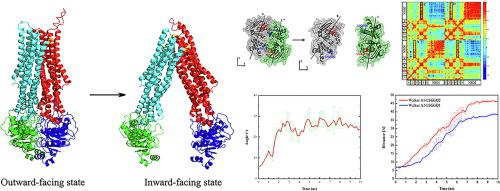
Human P-glycoprotein (P-gp) belongs to ATP-Binding Cassette (ABC) transporter, which can export varieties of anti-cancer drugs out of tumor cells, resulting in multidrug resistance (MDR) of tumor cells. We utilized targeted molecular dynamics simulation to explore the allosteric transition of human P-gp from outward- to inward-facing state. The results show that NBDs experience translational and rotational movement, caused by the successive hydrolysis of ATP molecules. The reorientation of the TMDs begins with the closing of the extracellular gate, followed by the opening of the cytoplasmic gate. The opening delay of the cytoplasmic gate is due to the extensive electrostatic interactions here. The sequence conservation analysis reveals that charged residues in TM3, 4 are highly conserved for achieving the unidirectional transport property. Our results give detailed movement and energy analyses to the reverse allosteric process of human P-gp and are helpful to the understanding of the working mechanism of human P-gp.
中文翻译:

从外向状态向内向人类P-糖蛋白变构转变的分子洞察
人P糖蛋白(P-gp)属于ATP结合盒(ABC)转运蛋白,可以将多种抗癌药物从肿瘤细胞中输出,从而导致肿瘤细胞产生多药耐药性(MDR)。我们利用有针对性的分子动力学模拟来探索人类P-gp从外向向内的变构转变。结果表明,NBD经历了由ATP分子的连续水解引起的平移和旋转运动。TMD的重新定向始于细胞外门的关闭,然后是细胞质门的打开。细胞质门的打开延迟是由于此处广泛的静电相互作用。序列保守性分析表明,TM3、4中带电的残基高度保守,可实现单向转运特性。

























 京公网安备 11010802027423号
京公网安备 11010802027423号