当前位置:
X-MOL 学术
›
Stem Cells Transl. Med.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
A pre-investigational new drug study of lung spheroid cell therapy for treating pulmonary fibrosis.
STEM CELLS Translational Medicine ( IF 6 ) Pub Date : 2020-04-18 , DOI: 10.1002/sctm.19-0167 Jhon Cores 1, 2 , Phuong-Uyen C Dinh 2 , Taylor Hensley 2 , Kenneth B Adler 2 , Leonard J Lobo 3 , Ke Cheng 1, 2, 4
STEM CELLS Translational Medicine ( IF 6 ) Pub Date : 2020-04-18 , DOI: 10.1002/sctm.19-0167 Jhon Cores 1, 2 , Phuong-Uyen C Dinh 2 , Taylor Hensley 2 , Kenneth B Adler 2 , Leonard J Lobo 3 , Ke Cheng 1, 2, 4
Affiliation
|
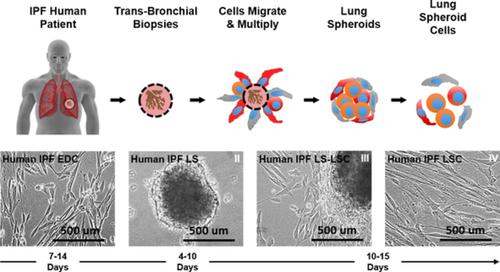
Idiopathic pulmonary fibrosis is a lethal interstitial lung disease with unknown etiology, no cure, and few treatment options. Herein, a therapy option is presented that makes use of a heterogeneous population of lung cells, including progenitor cells and supporting cells lines, cultured in adherent and suspension conditions, the latter of which induces spontaneous spheroid formation. Within these spheroids, progenitor marker expression is augmented. The cells, called lung spheroid cells, are isolated from fibrotic lungs, expanded, and delivered in single cell suspensions into rat models of pulmonary fibrosis via tail‐vein injections. Two bleomycin‐induced fibrotic rat models are used; a syngeneic Wistar‐Kyoto rat model, treated with syngeneic cells, and a xenogeneic nude rat model, treated with human cells. The first objective was to study the differences in fibrotic progression in the two rat models after bleomycin injury. Nude rat fibrosis formed quickly and extended for 30 days with no self‐resolution. Wistar‐Kyoto rat fibrosis was more gradual and began to decrease in severity between days 14 and 30. The second goal was to find the minimum effective dose of cells that demonstrated safe and effective therapeutic value. The resultant minimum effective therapeutic dose, acquired from the nude rat model, was 3 × 106 human cells. Histological analysis revealed no evidence of tumorigenicity, increased local immunological activity in the lungs, or an increase in liver enzyme production. These data demonstrate the safety and efficacy of lung spheroid cells in their application as therapeutic agents for pulmonary fibrosis, as well as their potential for clinical translation.
中文翻译:

肺球细胞疗法治疗肺纤维化的研究前新药研究。
特发性肺纤维化是一种致死性间质性肺疾病,病因不明,无法治愈,治疗选择很少。在此,提出了一种治疗方案,该方案利用了在贴壁和悬浮条件下培养的异种肺细胞群体,包括祖细胞和支持细胞系,后者可诱导自发球体形成。在这些球体中,祖细胞标记表达增加。这些细胞被称为肺球形细胞,从纤维化的肺中分离出来,扩增,并以单细胞悬液的形式通过尾静脉注射递送到大鼠肺纤维化模型中。使用了两种博来霉素诱导的纤维化大鼠模型。用同系细胞治疗的同基因Wistar‐Kyoto大鼠模型和用人细胞治疗的异种裸鼠模型。第一个目的是研究博来霉素损伤后两种大鼠模型中纤维化进程的差异。裸鼠纤维化迅速形成,并持续30天,没有自我分辨。Wistar-Kyoto大鼠纤维化更为缓慢,并在14天到30天之间开始减轻程度。第二个目标是找到证明安全有效的细胞最小有效剂量。从裸鼠模型获得的最小有效治疗剂量为3×10 第二个目标是寻找证明安全有效治疗价值的最小细胞有效剂量。从裸鼠模型获得的最小有效治疗剂量为3×10 第二个目标是寻找证明安全有效治疗价值的最小细胞有效剂量。从裸鼠模型获得的最小有效治疗剂量为3×106个人体细胞。组织学分析未发现致瘤性,肺部局部免疫活性增加或肝酶产生增加的证据。这些数据证明了肺球形细胞作为肺纤维化治疗剂的安全性和有效性,以及它们在临床上的翻译潜力。
更新日期:2020-04-18
中文翻译:

肺球细胞疗法治疗肺纤维化的研究前新药研究。
特发性肺纤维化是一种致死性间质性肺疾病,病因不明,无法治愈,治疗选择很少。在此,提出了一种治疗方案,该方案利用了在贴壁和悬浮条件下培养的异种肺细胞群体,包括祖细胞和支持细胞系,后者可诱导自发球体形成。在这些球体中,祖细胞标记表达增加。这些细胞被称为肺球形细胞,从纤维化的肺中分离出来,扩增,并以单细胞悬液的形式通过尾静脉注射递送到大鼠肺纤维化模型中。使用了两种博来霉素诱导的纤维化大鼠模型。用同系细胞治疗的同基因Wistar‐Kyoto大鼠模型和用人细胞治疗的异种裸鼠模型。第一个目的是研究博来霉素损伤后两种大鼠模型中纤维化进程的差异。裸鼠纤维化迅速形成,并持续30天,没有自我分辨。Wistar-Kyoto大鼠纤维化更为缓慢,并在14天到30天之间开始减轻程度。第二个目标是找到证明安全有效的细胞最小有效剂量。从裸鼠模型获得的最小有效治疗剂量为3×10 第二个目标是寻找证明安全有效治疗价值的最小细胞有效剂量。从裸鼠模型获得的最小有效治疗剂量为3×10 第二个目标是寻找证明安全有效治疗价值的最小细胞有效剂量。从裸鼠模型获得的最小有效治疗剂量为3×106个人体细胞。组织学分析未发现致瘤性,肺部局部免疫活性增加或肝酶产生增加的证据。这些数据证明了肺球形细胞作为肺纤维化治疗剂的安全性和有效性,以及它们在临床上的翻译潜力。

























 京公网安备 11010802027423号
京公网安备 11010802027423号