当前位置:
X-MOL 学术
›
Propellants Explos. Pyrotech.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
The Effects of Aniline Stabilizers on Nitrocellulose Based on Isothermal Thermal Decomposition
Propellants, Explosives, Pyrotechnics ( IF 1.8 ) Pub Date : 2020-02-27 , DOI: 10.1002/prep.201900390 Liqiong Luo 1 , Bo Jin 1 , Zuohu Chai 1 , Qiong Huang 1 , Shijin Chu 1 , Rufang Peng 1
Propellants, Explosives, Pyrotechnics ( IF 1.8 ) Pub Date : 2020-02-27 , DOI: 10.1002/prep.201900390 Liqiong Luo 1 , Bo Jin 1 , Zuohu Chai 1 , Qiong Huang 1 , Shijin Chu 1 , Rufang Peng 1
Affiliation
|
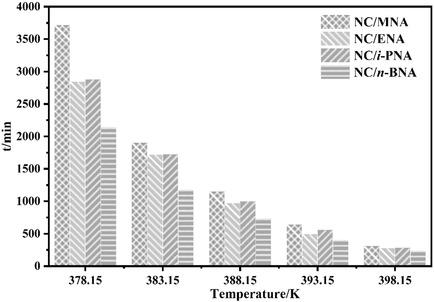
The effects of various aniline stabilizers on the thermal stability of nitrocellulose (NC) were investigated via isothermal thermal decomposition dynamics. The pressure‐time curves of NC/stabilizer composites were found to have a distinct inflection point, which was not observed on the pressure‐time curves of NC. The effects of stabilizers on the isothermal thermal decomposition kinetics of NC were evaluated using Arrhenius equation and model‐fitting method, and the effect of stabilizers on the storage time of NC was estimated using Berthelot equation. Results showed that the activation energy of NC/MNA, NC/ENA, NC/i ‐PNA, and NC/n ‐BNA before the inflection point were 162.9, 153.3, 147.4, and 150.6 kJ mol−1, respectively, which were significantly increased compared with that of pure NC (144.7 kJ mol−1). Furthermore, the time required for the decomposition extent of NC/MNA, NC/ENA, NC/i ‐PNA, and NC/n ‐BNA to reach 0.1 % were 24.9, 11.2, 16.7, and 11.2 years, respectively, at 298.15 K, which was longer than that required of NC (10.6 years). UV‐vis revealed that the stabilizers were almost consumed when the NC/stabilizer thermal decomposition pressure‐time curve reached the inflection point. Based on the above results, the stability order was MNA>i ‐PNA>ENA>n ‐BNA as the criterion for time of the inflection point.
中文翻译:

等温热分解对苯胺稳定剂对硝化纤维素的影响
通过等温热分解动力学研究了各种苯胺稳定剂对硝化纤维素(NC)热稳定性的影响。发现NC /稳定剂复合材料的压力-时间曲线具有明显的拐点,而在NC的压力-时间曲线上没有观察到。使用Arrhenius方程和模型拟合方法评估了稳定剂对NC的等温热分解动力学的影响,并使用Berthelot方程估算了稳定剂对NC的储存时间的影响。结果显示拐点之前的NC / MNA,NC / ENA,NC / i- PNA和NC / n- BNA的活化能为162.9、153.3、147.4和150.6 kJ mol -1分别比纯NC(144.7 kJ mol -1)显着增加。此外,NC / MNA,NC / ENA,NC / i- PNA和NC / n- BNA的分解程度达到0.1%所需的时间分别为298.15 K的24.9、11.2、16.7和11.2年。 ,比NC要求的时间长(10.6年)。UV-vis显示,当NC /稳定剂热分解压力-时间曲线达到拐点时,稳定剂几乎被消耗掉了。根据以上结果,稳定性顺序为MNA> i- PNA> ENA> n- BNA作为拐点时间的标准。
更新日期:2020-02-27
中文翻译:

等温热分解对苯胺稳定剂对硝化纤维素的影响
通过等温热分解动力学研究了各种苯胺稳定剂对硝化纤维素(NC)热稳定性的影响。发现NC /稳定剂复合材料的压力-时间曲线具有明显的拐点,而在NC的压力-时间曲线上没有观察到。使用Arrhenius方程和模型拟合方法评估了稳定剂对NC的等温热分解动力学的影响,并使用Berthelot方程估算了稳定剂对NC的储存时间的影响。结果显示拐点之前的NC / MNA,NC / ENA,NC / i- PNA和NC / n- BNA的活化能为162.9、153.3、147.4和150.6 kJ mol -1分别比纯NC(144.7 kJ mol -1)显着增加。此外,NC / MNA,NC / ENA,NC / i- PNA和NC / n- BNA的分解程度达到0.1%所需的时间分别为298.15 K的24.9、11.2、16.7和11.2年。 ,比NC要求的时间长(10.6年)。UV-vis显示,当NC /稳定剂热分解压力-时间曲线达到拐点时,稳定剂几乎被消耗掉了。根据以上结果,稳定性顺序为MNA> i- PNA> ENA> n- BNA作为拐点时间的标准。

























 京公网安备 11010802027423号
京公网安备 11010802027423号