当前位置:
X-MOL 学术
›
Biomater. Sci.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Delivery of dual miRNA through CD44-targeted mesoporous silica nanoparticles for enhanced and effective triple-negative breast cancer therapy.
Biomaterials Science ( IF 6.6 ) Pub Date : 2020-04-22 , DOI: 10.1039/d0bm00015a Manisha Ahir 1 , Priyanka Upadhyay , Avijit Ghosh , Sushmita Sarker , Saurav Bhattacharya , Payal Gupta , Swatilekha Ghosh , Sreya Chattopadhyay , Arghya Adhikary
Biomaterials Science ( IF 6.6 ) Pub Date : 2020-04-22 , DOI: 10.1039/d0bm00015a Manisha Ahir 1 , Priyanka Upadhyay , Avijit Ghosh , Sushmita Sarker , Saurav Bhattacharya , Payal Gupta , Swatilekha Ghosh , Sreya Chattopadhyay , Arghya Adhikary
Affiliation
|
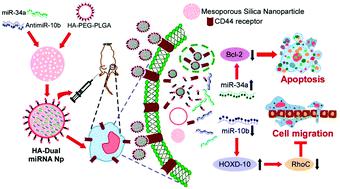
The development of new therapeutic strategies to target triple-negative breast cancer (TNBC) is in much demand to overcome the roadblocks associated with the existing treatment procedures. In this regard, therapies targeting the CD44 receptor have drawn attention for more than a decade. MicroRNAs (miRNAs) modulate post-transcriptional gene regulation and thus, the correction of specific miRNA alterations using miRNA mimics or antagomiRs is an emerging strategy to normalize the genetic regulation in the tumor microenvironment. It has been acknowledged that miR-34a is downregulated and miR-10b is upregulated in TNBC, which promotes tumorigenesis and metastatic dissemination. However, there are a few barriers related to miRNA delivery. Herein, we have introduced tailored mesoporous silica nanoparticles (MSNs) for the co-delivery of miR-34a-mimic and antisense-miR-10b. MSN was functionalized with a cationic basic side chain and then loaded with the dual combination to overexpress miR-34a and downregulate miR-10b simultaneously. Finally, the loaded MSNs were coated with an hyaluronic acid-appended PEG-PLGA polymer for specific targeting. The cellular uptake, release profile, and subsequent effect in TNBC cells were evaluated. In vitro and in vivo studies demonstrated high specificity in TNBC tumor targeting, leading to efficient tumor growth inhibition as well as the retardation of metastasis, which affirmed the clinical application potential of the system.
中文翻译:

通过靶向CD44的中孔二氧化硅纳米颗粒递送双重miRNA,以增强和有效治疗三阴性乳腺癌。
迫切需要开发针对三阴性乳腺癌(TNBC)的新治疗策略,以克服与现有治疗程序相关的障碍。在这方面,靶向CD44受体的疗法已经引起了十多年的关注。MicroRNA(miRNA)调节转录后的基因调控,因此,使用miRNA模仿物或antagomiRs纠正特定miRNA的改变是使肿瘤微环境中的基因调控正常化的新兴策略。公认的是,TNBC中miR-34a被下调,miR-10b被上调,这促进了肿瘤发生和转移扩散。但是,有一些与miRNA传递有关的障碍。在这里 我们已经为miR-34a-mimic和反义miR-10b共同提供了量身定制的介孔二氧化硅纳米颗粒(MSN)。MSN用阳离子基本侧链官能化,然后装载双重组合以过表达miR-34a和同时下调miR-10b。最后,用透明质酸键合的PEG-PLGA聚合物包被的MSN进行特异性靶向。评估了在TNBC细胞中的细胞吸收,释放情况和后续作用。体外和体内研究表明,在TNBC肿瘤靶向中具有高度特异性,从而导致有效的肿瘤生长抑制以及转移的延迟,这证实了该系统的临床应用潜力。MSN用阳离子基本侧链功能化,然后加载双重组合以过表达miR-34a和同时下调miR-10b。最后,用透明质酸键合的PEG-PLGA聚合物包被的MSN进行特异性靶向。评估了在TNBC细胞中的细胞吸收,释放情况和后续作用。体外和体内研究表明,在TNBC肿瘤靶向中具有高度特异性,从而导致有效的肿瘤生长抑制以及转移的延迟,这证实了该系统的临床应用潜力。MSN用阳离子基本侧链官能化,然后装载双重组合以过表达miR-34a和同时下调miR-10b。最后,将负载透明质酸的MSN涂上透明质酸附着的PEG-PLGA聚合物,以实现特异性靶向。评估了在TNBC细胞中的细胞吸收,释放情况和后续作用。体外和体内研究表明,在TNBC肿瘤靶向中具有高度特异性,从而导致有效的肿瘤生长抑制以及转移的延迟,这证实了该系统的临床应用潜力。
更新日期:2020-04-06
中文翻译:

通过靶向CD44的中孔二氧化硅纳米颗粒递送双重miRNA,以增强和有效治疗三阴性乳腺癌。
迫切需要开发针对三阴性乳腺癌(TNBC)的新治疗策略,以克服与现有治疗程序相关的障碍。在这方面,靶向CD44受体的疗法已经引起了十多年的关注。MicroRNA(miRNA)调节转录后的基因调控,因此,使用miRNA模仿物或antagomiRs纠正特定miRNA的改变是使肿瘤微环境中的基因调控正常化的新兴策略。公认的是,TNBC中miR-34a被下调,miR-10b被上调,这促进了肿瘤发生和转移扩散。但是,有一些与miRNA传递有关的障碍。在这里 我们已经为miR-34a-mimic和反义miR-10b共同提供了量身定制的介孔二氧化硅纳米颗粒(MSN)。MSN用阳离子基本侧链官能化,然后装载双重组合以过表达miR-34a和同时下调miR-10b。最后,用透明质酸键合的PEG-PLGA聚合物包被的MSN进行特异性靶向。评估了在TNBC细胞中的细胞吸收,释放情况和后续作用。体外和体内研究表明,在TNBC肿瘤靶向中具有高度特异性,从而导致有效的肿瘤生长抑制以及转移的延迟,这证实了该系统的临床应用潜力。MSN用阳离子基本侧链功能化,然后加载双重组合以过表达miR-34a和同时下调miR-10b。最后,用透明质酸键合的PEG-PLGA聚合物包被的MSN进行特异性靶向。评估了在TNBC细胞中的细胞吸收,释放情况和后续作用。体外和体内研究表明,在TNBC肿瘤靶向中具有高度特异性,从而导致有效的肿瘤生长抑制以及转移的延迟,这证实了该系统的临床应用潜力。MSN用阳离子基本侧链官能化,然后装载双重组合以过表达miR-34a和同时下调miR-10b。最后,将负载透明质酸的MSN涂上透明质酸附着的PEG-PLGA聚合物,以实现特异性靶向。评估了在TNBC细胞中的细胞吸收,释放情况和后续作用。体外和体内研究表明,在TNBC肿瘤靶向中具有高度特异性,从而导致有效的肿瘤生长抑制以及转移的延迟,这证实了该系统的临床应用潜力。

























 京公网安备 11010802027423号
京公网安备 11010802027423号