Marine Chemistry ( IF 3 ) Pub Date : 2020-03-26 , DOI: 10.1016/j.marchem.2020.103791 Liang Xue , Wei-Jun Cai
|
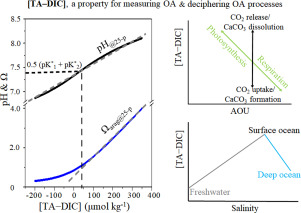
Ocean acidification (OA) defined as the decline of ocean pH and calcium carbonate saturation state (Ω) as a result of ocean uptake of CO2 from the atmosphere may have considerable negative impacts on global marine organisms and may substantially modify ocean biogeochemistry. However, as changes of pH and Ω are not conservative or linear with respect to ocean physical processes (e.g., mixing, temperature and pressure changes), the influences of anthropogenic CO2 uptake and ocean biogeochemical processes on OA rates cannot be easily identified. Here, we examine whether a composite property [TA–DIC] or the difference between total alkalinity (TA) and dissolved inorganic carbon (DIC), which is conservative to ocean mixing and is not sensitive to temperature and pressure changes, can be used for measuring OA rates and deciphering the underlying OA mechanisms in the global ocean as it in surface waters of several regional oceans. Based on Global Ocean Data Analysis Project Version 2 (GLODAPv2), we demonstrate using this property for measuring OA rates can be applied on a global ocean scale, except at low salinity e.g., < 20 and when [TA–DIC] is <~50 μmol kg−1, where the relationships of [TA–DIC] with pH and/or Ω are nonlinear. However, there are almost no limitations when using this property for deciphering the underlying OA mechanisms since the change of [TA–DIC] with time is relatively small on OA timescales of decades or more. Using [TA–DIC], we can readily quantify the influences from freshwater inputs and upwelling on OA rates based on a two end-member mixing model. More importantly, through the Redfield ratio and apparent oxygen utilization, we can directly link biological influences to OA rates and conveniently quantify the biological modulation on OA rates. Therefore, we argue that using [TA–DIC] as a proxy for OA would provide a simple but powerful way of deciphering acidification mechanisms and predicting future development of acidification.
中文翻译:

总碱度减去溶解的无机碳可解释海洋酸化机制
海洋酸化(OA)定义为海洋从大气中吸收CO 2导致海洋pH下降和碳酸钙饱和状态(Ω)下降,可能对全球海洋生物产生相当大的负面影响,并可能大大改变海洋生物地球化学。但是,由于pH和Ω的变化相对于海洋物理过程(例如,混合,温度和压力变化)不是保守的或线性的,因此人为CO 2的影响OA速率的吸收和海洋生物地球化学过程不容易确定。在这里,我们检查是否可以将复合性质[TA–DIC]或总碱度(TA)与溶解的无机碳(DIC)之间的差异(对海洋混合而言是保守的并且对温度和压力变化不敏感)用于测量OA速率,并解释全球海洋在几个区域海洋的表层水中的潜在OA机制。基于全球海洋数据分析项目第2版(GLODAPv2),我们证明了使用此属性测量OA速率可以在全球海洋规模上应用,但在低盐度(例如<20和[TA–DIC] <〜50)时除外μmol千克-1,其中[TA–DIC]与pH和/或Ω的关系是非线性的。但是,使用此属性来解密潜在的OA机制时几乎没有限制,因为在几十年甚至更长的OA时间尺度上,[TA–DIC]随时间的变化相对较小。使用[TA–DIC],我们可以基于两个末端成员混合模型轻松量化淡水输入和上升对OA速率的影响。更重要的是,通过雷德菲尔德比和表观氧气利用率,我们可以将生物学影响与OA速率直接联系起来,并方便地量化对OA速率的生物调节。因此,我们认为使用[TA–DIC]作为OA的代理将提供一种简单而有效的方法来破译酸化机制并预测酸化的未来发展。


























 京公网安备 11010802027423号
京公网安备 11010802027423号