当前位置:
X-MOL 学术
›
J. Pharmaceut. Biomed. Anal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Novel method for fast trabectedin quantification using hydrophilic interaction liquid chromatography and tandem mass spectrometry for human pharmacokinetic studies.
Journal of Pharmaceutical and Biomedical Analysis ( IF 3.4 ) Pub Date : 2020-03-17 , DOI: 10.1016/j.jpba.2020.113261 Emanuela Di Gregorio 1 , Gianmaria Miolo 2 , Agostino Steffan 3 , Giuseppe Corona 3
Journal of Pharmaceutical and Biomedical Analysis ( IF 3.4 ) Pub Date : 2020-03-17 , DOI: 10.1016/j.jpba.2020.113261 Emanuela Di Gregorio 1 , Gianmaria Miolo 2 , Agostino Steffan 3 , Giuseppe Corona 3
Affiliation
|
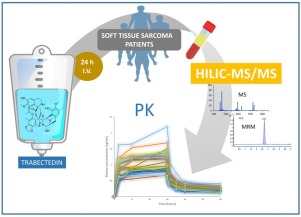
Few time-consuming bioanalytical methods are currently available for trabectedin quantification in clinical investigations. Here we present a novel, fast and sensitive method for trabectedin determination in human plasma based on hydrophilic interaction liquid chromatography and tandem mass spectrometry (HILIC-MS/MS). Plasma samples are treated with acetonitrile-0.1 % formic acid and the solvent extract is directly injected into an Acquity BEH Amide column (2.1 × 100 mm, 1.7 μm) operating in HILIC mode at 0.2 mL/min with 80:20 acetonitrile-0.1 % formic acid in water. The analyte is separated by an organic solvent gradient and quantified by an Agilent Ultivo triple quadrupole mass spectrometer operating in multiple reaction monitoring (MRM) mode. The quantitative MRM transitions were m/z 762→234 and m/z 765→234 for trabectedin and its d3-labeled derivative, respectively. The lower limit of quantification (LLOQ) was 0.01 ng/mL and the assay was linear up to 2.5 ng/mL. The intra- and inter-day relative error ranged from 1.19 % to 8.52 %, while the relative standard deviation was less than 12.35 %. The method was used to determine the pharmacokinetic profiles of trabectedin in 26 patients with soft tissue sarcoma, showing that this new HILIC-MS/MS method is suitable for use in clinical research.
中文翻译:

使用亲水相互作用液相色谱和串联质谱进行人体药物动力学研究的快速trabectedin定量新方法。
目前很少有耗时的生物分析方法可用于临床研究中trabectedin的定量分析。在这里,我们介绍了一种基于亲水相互作用液相色谱和串联质谱(HILIC-MS / MS)的新型,快速,灵敏的人体血浆曲布汀测定方法。血浆样品用0.1%的乙腈溶液处理,并将溶剂萃取液直接注入以HILIC模式运行的Acquity BEH Amide色谱柱(2.1×100 mm,1.7μm),以80:20的乙腈-0.1%在0.2 mL / min下运行水中有甲酸。分析物通过有机溶剂梯度分离,并通过在多反应监测(MRM)模式下运行的Agilent Ultivo三重四极杆质谱仪进行定量。trabectedin及其d3标记衍生物的定量MRM跃迁分别为m / z 762→234和m / z 765→234。定量下限(LLOQ)为0.01 ng / mL,线性分析最高为2.5 ng / mL。日内和日间相对误差在1.19%至8.52%范围内,而相对标准偏差小于12.35%。该方法用于测定曲贝汀在26例软组织肉瘤患者中的药代动力学,表明该新的HILIC-MS / MS方法适用于临床研究。
更新日期:2020-03-19
中文翻译:

使用亲水相互作用液相色谱和串联质谱进行人体药物动力学研究的快速trabectedin定量新方法。
目前很少有耗时的生物分析方法可用于临床研究中trabectedin的定量分析。在这里,我们介绍了一种基于亲水相互作用液相色谱和串联质谱(HILIC-MS / MS)的新型,快速,灵敏的人体血浆曲布汀测定方法。血浆样品用0.1%的乙腈溶液处理,并将溶剂萃取液直接注入以HILIC模式运行的Acquity BEH Amide色谱柱(2.1×100 mm,1.7μm),以80:20的乙腈-0.1%在0.2 mL / min下运行水中有甲酸。分析物通过有机溶剂梯度分离,并通过在多反应监测(MRM)模式下运行的Agilent Ultivo三重四极杆质谱仪进行定量。trabectedin及其d3标记衍生物的定量MRM跃迁分别为m / z 762→234和m / z 765→234。定量下限(LLOQ)为0.01 ng / mL,线性分析最高为2.5 ng / mL。日内和日间相对误差在1.19%至8.52%范围内,而相对标准偏差小于12.35%。该方法用于测定曲贝汀在26例软组织肉瘤患者中的药代动力学,表明该新的HILIC-MS / MS方法适用于临床研究。

























 京公网安备 11010802027423号
京公网安备 11010802027423号