Communications Biology ( IF 5.9 ) Pub Date : 2020-03-13 , DOI: 10.1038/s42003-020-0855-y Hau-Ming Jan , Yi-Chi Chen , Tsai-Chen Yang , Lih-Lih Ong , Chia-Chen Chang , Sasikala Muthusamy , Andualem Bahiru Abera , Ming-Shiang Wu , Jacquelyn Gervay-Hague , Kwok-Kong Tony Mong , Chun-Hung Lin
|
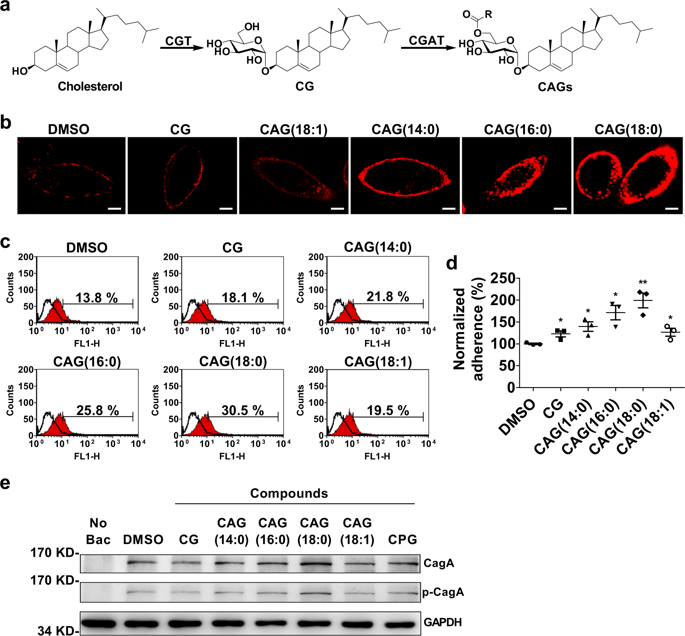
Helicobacter pylori, the most common etiologic agent of gastric diseases including gastric cancer, is auxotrophic for cholesterol and has to hijack it from gastric epithelia. Upon uptake, the bacteria convert cholesterol to cholesteryl 6′-O-acyl-α-D-glucopyranoside (CAG) to promote lipid raft clustering in the host cell membranes. However, how CAG appears in the host to exert the pathogenesis still remains ambiguous. Herein we identified hp0499 to be the gene of cholesteryl α-D-glucopyranoside acyltransferase (CGAT). Together with cholesteryl glucosyltransferase (catalyzing the prior step), CGAT is secreted via outer membrane vesicles to the host cells for direct synthesis of CAG. This significantly enhances lipid rafts clustering, gathers adhesion molecules (including Lewis antigens and integrins α5, β1), and promotes more bacterial adhesion. Furthermore, the clinically used drug amiodarone was shown as a potent inhibitor of CGAT to effectively reduce the bacterial adhesion, indicating that CGAT is a potential target of therapeutic intervention.
中文翻译:

胆固醇α-D-葡萄糖苷6-酰基转移酶增强幽门螺杆菌对胃上皮的粘附
幽门螺杆菌(Helicobacter pylori)是包括胃癌在内的胃病最常见的病因,它对胆固醇具有营养缺陷,必须从胃上皮中劫持。摄取后,细菌将胆固醇转化为胆固醇基6'- O-酰基-α-D-吡喃葡萄糖苷(CAG),以促进脂质筏在宿主细胞膜中聚集。但是,CAG如何在宿主中出现以发挥发病机理仍然不清楚。在此我们确定了hp0499是胆固醇α-D-吡喃葡萄糖苷酰基转移酶(CGAT)的基因。CGAT与胆固醇基葡萄糖基转移酶(催化上一步骤)一起通过外膜囊泡分泌到宿主细胞中,以直接合成CAG。这显着增强了脂筏的聚集,聚集了粘附分子(包括Lewis抗原和整联蛋白α5,β1),并促进了更多细菌的粘附。此外,临床上使用的胺碘酮已被证明是有效抑制细菌粘附的有效CGAT抑制剂,表明CGAT是治疗干预的潜在靶标。

























 京公网安备 11010802027423号
京公网安备 11010802027423号