当前位置:
X-MOL 学术
›
J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
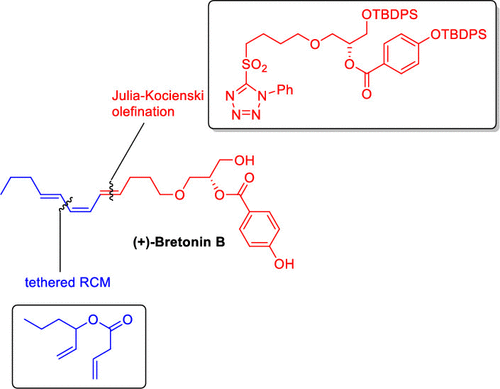
Stereoselective Synthesis of Conjugated Polyenes Based on Tethered Olefin Metathesis and Carbonyl Olefination: Application to the Total Synthesis of (+)-Bretonin B
The Journal of Organic Chemistry ( IF 3.6 ) Pub Date : 2020-03-18 , DOI: 10.1021/acs.joc.0c00446 Kajsa Lood 1 , Bernd Schmidt 1
The Journal of Organic Chemistry ( IF 3.6 ) Pub Date : 2020-03-18 , DOI: 10.1021/acs.joc.0c00446 Kajsa Lood 1 , Bernd Schmidt 1
Affiliation
|
The combination of a highly stereoselective tethered olefin metathesis reaction and a Julia–Kocienski olefination is presented as a strategy for the synthesis of conjugated polyenes with at least one Z-configured C═C bond. The strategy is exemplified by the synthesis of the marine natural product (+)-bretonin B.
中文翻译:

基于链状烯烃复分解和羰基烯烃化的共轭多烯立体选择性合成:在(+)-Bretonin B的全合成中的应用
高度立体选择性束缚的烯烃复分解反应和Julia-Kocienski烯烃化的结合被提出作为合成具有至少一个Z构型的C═C键的多烯的策略。该策略以海洋天然产物(+)-白蛋白B的合成为例。
更新日期:2020-03-19
中文翻译:

基于链状烯烃复分解和羰基烯烃化的共轭多烯立体选择性合成:在(+)-Bretonin B的全合成中的应用
高度立体选择性束缚的烯烃复分解反应和Julia-Kocienski烯烃化的结合被提出作为合成具有至少一个Z构型的C═C键的多烯的策略。该策略以海洋天然产物(+)-白蛋白B的合成为例。

























 京公网安备 11010802027423号
京公网安备 11010802027423号