当前位置:
X-MOL 学术
›
Green Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Catalytic transfer hydrogenation of maleic acid with stoichiometric amounts of formic acid in aqueous phase: paving the way for more sustainable succinic acid production
Green Chemistry ( IF 9.8 ) Pub Date : 2020/02/25 , DOI: 10.1039/c9gc04221k M. López Granados 1, 2, 3, 4 , J. Moreno 4, 5, 6, 7 , A. C. Alba-Rubio 8, 9, 10, 11 , J. Iglesias 4, 5, 6, 7 , D. Martín Alonso 11, 12, 13, 14, 15 , R. Mariscal 1, 2, 3, 4
Green Chemistry ( IF 9.8 ) Pub Date : 2020/02/25 , DOI: 10.1039/c9gc04221k M. López Granados 1, 2, 3, 4 , J. Moreno 4, 5, 6, 7 , A. C. Alba-Rubio 8, 9, 10, 11 , J. Iglesias 4, 5, 6, 7 , D. Martín Alonso 11, 12, 13, 14, 15 , R. Mariscal 1, 2, 3, 4
Affiliation
|
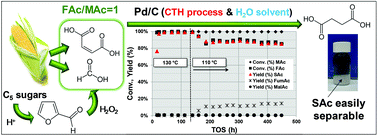
The aqueous phase hydrogenation of maleic acid (MAc) to succinic acid (SAc) is demonstrated in the absence of any organic solvent and using stoichiometric amount of formic acid (FAc) as source of H2. Among the different noble metals (Pd, Au, Ru, Pt and Rh) and supports investigated (γ-Al2O3, TiO2, CeO2, ZrO2, WO3, CeZrO4, carbon, nicanite, SiO2 and TS-1), Pd/C was identified as the best catalyst. We observe that the undesirable formation of malic acid (MalAc) by hydration of MAc must be prevented. The transformation of MAc to SAc with negligible formation of MalAc is possible by using relatively mild temperature (140–150 °C) and a high catalyst to MAc ratio (i.e. fixed bed continuous flow reactor). Using the carboxylate forms (disodium maleate and sodium formate) instead of the acids results in an increase of the reaction rate. In a fixed bed reactor under a continuous flow of 15 wt% of MAc at a WHSV = 12 h−1 (contact time = 5 min), at 150 °C, 10 bar of N2 and using a formic acid/maleic acid molar ratio = 1, a yield of SAc close to 98% was obtained, equivalent to a productivity of 1.87 g SAc per gcat per·h. Leaching of Pd was below 0.02 ppm. No deactivation was observed in long term experiments at 150 °C (ca. 730 h), although the characterization of the used catalyst by CO chemisorption and TEM and XPS studies showed certain sintering of Pd particles. Regarding the mechanism of the reaction, kinetic isotopic experiments using deuterated DCOOH indicated that the reaction must essentially proceed via catalytic transfer hydrogenation, formyl H of formic acid is involved in the rate determining step of the reaction. When using maleate and formate sodium salts, the second H needed for the reaction is supplied by the solvent (H2O molecules). A preliminary environmental assessment (Life Cycle Analysis, LCA) of this CTH approach indicates that for relevant environmental categories of the LCA (such as climate change and consumption of fossil resources) the CTH process is greener than conventional hydrogenation process; the benefits are even larger if biomass-derived FAc is involved.
中文翻译:

在水相中以化学计量的甲酸催化转移氢化马来酸:为更可持续的琥珀酸生产铺平道路
在不存在任何有机溶剂并且使用化学计量的甲酸(FAc)作为H 2的来源下,证明了马来酸(MAc)的水相氢化为琥珀酸(SAc)。在被调查的不同贵金属(钯,金,钌,铂和铑)和支撑件(在γ-Al 2 ö 3,的TiO 2,的CeO 2,的ZrO 2,WO 3,CeZrO 4,碳,nicanite的SiO 2和TS-1),Pd / C被认为是最好的催化剂。我们观察到,必须防止由于MAc的水合而形成不想要的苹果酸(MalAc)。通过使用相对温和的温度(140–150°C)和较高的催化剂与MAc比率(即固定床连续流反应器),可以将MAc转化为SAc,而形成的MalAc可以忽略不计。使用羧酸盐形式(马来酸二钠和甲酸钠)代替酸会导致反应速率增加。在固定床反应器中,在WHSV = 12 h -1(接触时间= 5分钟)下,在15 wt%MAc的连续流动下,在150°C下,使用10 bar N 2并使用甲酸/马来酸摩尔比率= 1,可获得接近98%的SAc产率,相当于每克1.87 g SAc的生产率每小时猫 Pd的浸出低于0.02 ppm。尽管在150°C(约730 h)的长期实验中未观察到失活,但是通过CO化学吸附以及TEM和XPS研究对使用过的催化剂进行了表征,结果表明对Pd颗粒进行了一定程度的烧结。关于反应的机理,使用氘代DCOOH的动力学同位素实验表明反应必须基本上通过催化转移氢化进行,甲酸的甲酰基H参与反应的速率确定步骤。当使用马来酸盐和甲酸钠盐时,反应所需的第二个H由溶剂(H 2O分子)。这种CTH方法的初步环境评估(生命周期分析,LCA)表明,对于LCA的相关环境类别(例如气候变化和化石资源的消耗),CTH过程比常规加氢过程更环保;如果涉及生物质衍生的FAc,则收益甚至更大。
更新日期:2020-03-24
中文翻译:

在水相中以化学计量的甲酸催化转移氢化马来酸:为更可持续的琥珀酸生产铺平道路
在不存在任何有机溶剂并且使用化学计量的甲酸(FAc)作为H 2的来源下,证明了马来酸(MAc)的水相氢化为琥珀酸(SAc)。在被调查的不同贵金属(钯,金,钌,铂和铑)和支撑件(在γ-Al 2 ö 3,的TiO 2,的CeO 2,的ZrO 2,WO 3,CeZrO 4,碳,nicanite的SiO 2和TS-1),Pd / C被认为是最好的催化剂。我们观察到,必须防止由于MAc的水合而形成不想要的苹果酸(MalAc)。通过使用相对温和的温度(140–150°C)和较高的催化剂与MAc比率(即固定床连续流反应器),可以将MAc转化为SAc,而形成的MalAc可以忽略不计。使用羧酸盐形式(马来酸二钠和甲酸钠)代替酸会导致反应速率增加。在固定床反应器中,在WHSV = 12 h -1(接触时间= 5分钟)下,在15 wt%MAc的连续流动下,在150°C下,使用10 bar N 2并使用甲酸/马来酸摩尔比率= 1,可获得接近98%的SAc产率,相当于每克1.87 g SAc的生产率每小时猫 Pd的浸出低于0.02 ppm。尽管在150°C(约730 h)的长期实验中未观察到失活,但是通过CO化学吸附以及TEM和XPS研究对使用过的催化剂进行了表征,结果表明对Pd颗粒进行了一定程度的烧结。关于反应的机理,使用氘代DCOOH的动力学同位素实验表明反应必须基本上通过催化转移氢化进行,甲酸的甲酰基H参与反应的速率确定步骤。当使用马来酸盐和甲酸钠盐时,反应所需的第二个H由溶剂(H 2O分子)。这种CTH方法的初步环境评估(生命周期分析,LCA)表明,对于LCA的相关环境类别(例如气候变化和化石资源的消耗),CTH过程比常规加氢过程更环保;如果涉及生物质衍生的FAc,则收益甚至更大。

























 京公网安备 11010802027423号
京公网安备 11010802027423号