Journal of Solid State Chemistry ( IF 3.3 ) Pub Date : 2020-02-22 , DOI: 10.1016/j.jssc.2020.121263 Anucha Koedtruad , Midori Amano Patino , Noriya Ichikawa , Daisuke Kan , Yuichi Shimakawa
|
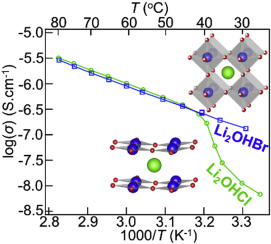
Crystal structures and ionic conductivity of antiperovskites Li2OHX (X = Cl, Br), which are potential solid electrolyte materials for all-solid-state Li-ion batteries, were investigated. Li2OHCl showed a Pmc21 orthorhombic structure at room temperature and changed to a cubic one between 27 and 37 °C. Li2OHBr, on the other hand, crystallized in a cubic structure and showed no phase transition in the measured temperature range. The cubic phases of both compounds showed dominant high Li+-ion conductivity, while the orthorhombic phase of Li2OHCl exhibited reduced ionic conductivity. The Li+-ion order in the orthorhombic Li2OHCl makes the crystal structure highly anisotropic and two dimensional, which disturbs the hopping of the Li+ ions.
中文翻译:

Li 2 OHX(X = Cl,Br)抗钙钛矿的晶体结构和离子电导率
研究了作为全固态锂离子电池潜在的固体电解质材料的抗钙钛矿Li 2 OHX(X = Cl,Br)的晶体结构和离子电导率。Li 2 OHCl在室温下显示Pmc 2 1正交结构,并变为27至37°C之间的立方立方。另一方面,Li 2 OHBr在呈立方结构,在测得的温度范围内无相变。两种化合物的立方相均显示出较高的Li +离子电导率,而Li 2 OHCl的斜方晶相则显示出较低的离子电导率。正交晶的Li 2 OHCl中的Li +离子顺序使晶体结构具有高度的各向异性和二维性,这扰乱了Li +离子的跳跃。



























 京公网安备 11010802027423号
京公网安备 11010802027423号