Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Developing in vitro assays to transform gastrointestinal safety assessment: potential for microphysiological systems.
Lab on a Chip ( IF 6.1 ) Pub Date : 2020-02-21 , DOI: 10.1039/c9lc01107b Matthew F Peters 1 , Allison L Choy , Carmen Pin , Derek J Leishman , Annie Moisan , Lorna Ewart , Peggy J Guzzie-Peck , Radhakrishna Sura , Douglas A Keller , Clay W Scott , Kyle L Kolaja
Lab on a Chip ( IF 6.1 ) Pub Date : 2020-02-21 , DOI: 10.1039/c9lc01107b Matthew F Peters 1 , Allison L Choy , Carmen Pin , Derek J Leishman , Annie Moisan , Lorna Ewart , Peggy J Guzzie-Peck , Radhakrishna Sura , Douglas A Keller , Clay W Scott , Kyle L Kolaja
Affiliation
|
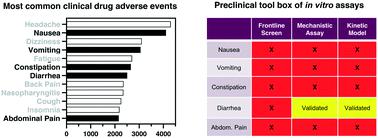
Drug-induced gastrointestinal toxicities (DI-GITs) are among the most common adverse events in clinical trials. High prevalence of DI-GIT has persisted among new drugs due in part to the lack of robust experimental tools to allow early detection or to guide optimization of safer molecules. Developing in vitro assays for the leading GI toxicities (nausea, vomiting, diarrhoea, constipation, and abdominal pain) will likely involve recapitulating complex physiological properties that require contributions from diverse cell/tissue types including epithelial, immune, microbiome, nerve, and muscle. While this stipulation may be beyond traditional 2D monocultures of intestinal cell lines, emerging 3D GI microtissues capture interactions between diverse cell and tissue types. These interactions give rise to microphysiologies fundamental to gut biology. For GI microtissues, organoid technology was the breakthrough that introduced intestinal stem cells with the capability of differentiating into each of the epithelial cell types and that self-organize into a multi-cellular tissue proxy with villus- and crypt-like domains. Recently, GI microtissues generated using miniaturized devices with microfluidic flow and cyclic peristaltic strain were shown to induce Caco2 cells to spontaneously differentiate into each of the principle intestinal epithelial cell types. Second generation models comprised of epithelial organoids or microtissues co-cultured with non-epithelial cell types can successfully reproduce cross-'tissue' functional interactions broadening the potential of these models to accurately study drug-induced toxicities. A new paradigm in which in vitro assays become an early part of GI safety assessment could be realized if microphysiological systems (MPS) are developed in alignment with drug-discovery needs. Herein, approaches for assessing GI toxicity of pharmaceuticals are reviewed and gaps are compared with capabilities of emerging GI microtissues (e.g., organoids, organ-on-a-chip, transwell systems) in order to provide perspective on the assay features needed for MPS models to be adopted for DI-GIT assessment.
中文翻译:

开发体外试验以改变胃肠道安全性评估:微生理系统的潜力。
药物引起的胃肠道毒性(DI-GITs)是临床试验中最常见的不良事件之一。DI-GIT 的高流行率在新药中一直存在,部分原因是缺乏强大的实验工具来允许早期检测或指导更安全分子的优化。开发针对主要 GI 毒性(恶心、呕吐、腹泻、便秘和腹痛)的体外测定可能涉及概括复杂的生理特性,这些特性需要来自不同细胞/组织类型(包括上皮、免疫、微生物组、神经和肌肉)的贡献。虽然这一规定可能超出了肠道细胞系的传统 2D 单一培养,但新兴的 3D GI 微组织捕获了不同细胞和组织类型之间的相互作用。这些相互作用产生了肠道生物学基础的微生理学。对于 GI 微组织,类器官技术是一项突破,它引入了具有分化成每种上皮细胞类型的能力的肠道干细胞,并且能够自我组织成具有绒毛和隐窝样结构域的多细胞组织代理。最近,使用具有微流体流动和循环蠕动应变的小型设备生成的 GI 微组织被证明可以诱导 Caco2 细胞自发分化成每种主要的肠上皮细胞类型。由上皮类器官或与非上皮细胞类型共培养的微组织组成的第二代模型可以成功地再现跨“组织”功能相互作用,从而拓宽了这些模型准确研究药物诱导毒性的潜力。如果根据药物发现需求开发微生理系统 (MPS),则可以实现体外检测成为 GI 安全性评估早期部分的新范例。本文回顾了评估药物 GI 毒性的方法,并将差距与新兴 GI 微组织(例如,类器官、芯片上的器官、transwell 系统)的能力进行比较,以提供对 MPS 模型所需的分析特征的看法用于 DI-GIT 评估。
更新日期:2020-02-21
中文翻译:

开发体外试验以改变胃肠道安全性评估:微生理系统的潜力。
药物引起的胃肠道毒性(DI-GITs)是临床试验中最常见的不良事件之一。DI-GIT 的高流行率在新药中一直存在,部分原因是缺乏强大的实验工具来允许早期检测或指导更安全分子的优化。开发针对主要 GI 毒性(恶心、呕吐、腹泻、便秘和腹痛)的体外测定可能涉及概括复杂的生理特性,这些特性需要来自不同细胞/组织类型(包括上皮、免疫、微生物组、神经和肌肉)的贡献。虽然这一规定可能超出了肠道细胞系的传统 2D 单一培养,但新兴的 3D GI 微组织捕获了不同细胞和组织类型之间的相互作用。这些相互作用产生了肠道生物学基础的微生理学。对于 GI 微组织,类器官技术是一项突破,它引入了具有分化成每种上皮细胞类型的能力的肠道干细胞,并且能够自我组织成具有绒毛和隐窝样结构域的多细胞组织代理。最近,使用具有微流体流动和循环蠕动应变的小型设备生成的 GI 微组织被证明可以诱导 Caco2 细胞自发分化成每种主要的肠上皮细胞类型。由上皮类器官或与非上皮细胞类型共培养的微组织组成的第二代模型可以成功地再现跨“组织”功能相互作用,从而拓宽了这些模型准确研究药物诱导毒性的潜力。如果根据药物发现需求开发微生理系统 (MPS),则可以实现体外检测成为 GI 安全性评估早期部分的新范例。本文回顾了评估药物 GI 毒性的方法,并将差距与新兴 GI 微组织(例如,类器官、芯片上的器官、transwell 系统)的能力进行比较,以提供对 MPS 模型所需的分析特征的看法用于 DI-GIT 评估。

























 京公网安备 11010802027423号
京公网安备 11010802027423号