Biosensors and Bioelectronics ( IF 12.6 ) Pub Date : 2020-02-19 , DOI: 10.1016/j.bios.2020.112113 Amin Hassanzadeh-Barforoushi , Majid Ebrahimi Warkiani , David Gallego-Ortega , Guozhen Liu , Tracie Barber
|
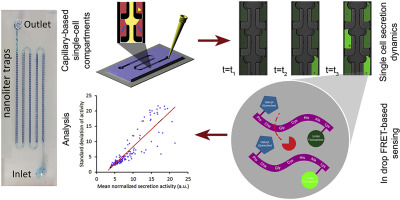
Cancer cells continuously secrete inflammatory biomolecules which play significant roles in disease progression and tumor metastasis toward secondary sites. Despite recent efforts to capture cancer cells' intercellular secretion heterogeneity using microfluidics, the challenges in operation of these systems as well as the complexity of designing a biosensing assay for long-term and real-time measurement of single cell secretions have become grand research barriers. Here, we present a new capillary-based microfluidic biosensing approach to easily and reliably capture ∼500 single cells inside isolated dead-end nanoliter compartments using simple pipette injection, and quantify their individual secretion dynamics at the single cell resolution over a long period of culture (∼16 h). We first present a detailed investigation of the fluid mechanics underlying the formation of nanoliter compartments in the microfluidic system. Based on the measurement of single cell capture efficiency, we employ a one-step FRET-based biosensor which monitors the single cancer cells' protease activity. The sensor reports the fluorescent signal as a product of amino acid chain cleavage and reduction in its quenching capability. Using the single cell protease secretion data, we identified modes of cell secretion dynamics in our cell sample. While most of the cells had low secretion levels, two other smaller and more aggressive secretion dynamics were cells aggressive cells with secretion modes that include sharp spikes or slow but progressive trend. The method presented here overcomes the difficulties associated with performing single cell secretion assays, enabling a feasible and reliable technique for high throughput measurement of metabolic activities in cancer cells.
中文翻译:

毛细管辅助微流生物传感平台可捕获纳升隔室中的单细胞分泌动力学
癌细胞不断分泌炎症性生物分子,这些分子在疾病进展和肿瘤向次要部位的转移中起重要作用。尽管最近进行了使用微流体技术来捕获癌细胞的细胞间分泌异质性的努力,但是这些系统在操作上的挑战以及设计用于长期和实时测量单细胞分泌物的生物传感测定的复杂性已成为研究的主要障碍。在这里,我们提出了一种新的基于毛细管的微流控生物传感方法,可通过简单的移液器轻松可靠地捕获隔离的死纳升隔室中的约500个单细胞,并在长期培养过程中以单细胞分辨率量化其个体分泌动力学(〜16小时)。我们首先提出对微流体系统中纳升隔室形成基础的流体力学的详细研究。基于对单细胞捕获效率的测量,我们采用了一种基于FRET的一步式生物传感器,该传感器可监视单个癌细胞的蛋白酶活性。传感器报告荧光信号是氨基酸链断裂和淬灭能力降低的产物。使用单细胞蛋白酶分泌数据,我们确定了我们细胞样品中细胞分泌动力学的模式。虽然大多数细胞的分泌水平较低,但另外两个较小且更具侵略性的分泌动力学是细胞 我们采用基于FRET的一步式生物传感器,该传感器可监测单个癌细胞的蛋白酶活性。传感器报告荧光信号是氨基酸链断裂和淬灭能力降低的产物。使用单细胞蛋白酶分泌数据,我们确定了我们细胞样品中细胞分泌动力学的模式。虽然大多数细胞的分泌水平较低,但另外两个较小且更具侵略性的分泌动力学是细胞 我们采用基于FRET的一步式生物传感器,该传感器可监测单个癌细胞的蛋白酶活性。传感器报告荧光信号是氨基酸链断裂和淬灭能力降低的产物。使用单细胞蛋白酶分泌数据,我们确定了我们细胞样品中细胞分泌动力学的模式。虽然大多数细胞的分泌水平较低,但另外两个较小且更具侵略性的分泌动力学是细胞侵袭性细胞分泌模式包括尖峰或缓慢但渐进的趋势。本文介绍的方法克服了与执行单细胞分泌测定相关的困难,从而使可行且可靠的技术能够高通量测量癌细胞中的代谢活性。

























 京公网安备 11010802027423号
京公网安备 11010802027423号