当前位置:
X-MOL 学术
›
JAMA Neurol.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Clinical Effects of the Self-administered Subcutaneous Complement Inhibitor Zilucoplan in Patients With Moderate to Severe Generalized Myasthenia Gravis: Results of a Phase 2 Randomized, Double-Blind, Placebo-Controlled, Multicenter Clinical Trial.
JAMA Neurology ( IF 29.0 ) Pub Date : 2020-05-01 , DOI: 10.1001/jamaneurol.2019.5125 James F Howard 1 , Richard J Nowak 2 , Gil I Wolfe 3 , Miriam L Freimer 4 , Tuan H Vu 5 , John L Hinton 6 , Michael Benatar 7 , Petra W Duda 8 , James E MacDougall 8 , Ramin Farzaneh-Far 8 , Henry J Kaminski 9 , , Richard Barohn 10 , Mazen Dimachkie 10 , Mamatha Pasnoor 10 , Constantine Farmakidis 10 , Tina Liu 10 , Samantha Colgan 10 , Michael G Benatar 7 , Tulio Bertorini 11 , Rekha Pillai 11 , Robert Henegar 11 , Mark Bromberg 12 , Summer Gibson 12 , Teresa Janecki 12 , Miriam Freimer 4 , Bakri Elsheikh 4 , Paige Matisak 4 , Angela Genge 13 , Amanda Guidon 14 , William David 14 , Ali A Habib 15 , Veena Mathew 15 , Tahseen Mozaffar 15 , John L Hinton 6 , William Hewitt 6 , Deborah Barnett 6 , Patricia Sullivan 6 , Doreen Ho 16 , James F Howard 1 , Rebecca E Traub 1 , Manisha Chopra 1 , Henry J Kaminski 9 , Radwa Aly 9 , Elham Bayat 9 , Mohammad Abu-Rub 9 , Shaida Khan 17 , Dale Lange 18 , Shara Holzberg 18 , Bhupendra Khatri 19 , Emily Lindman 19 , Tayo Olapo 19 , Lisa M Sershon 19 , Robert P Lisak 20 , Evanthia Bernitsas 20 , Kelly Jia 20 , Rabia Malik 21 , Tiffany D Lewis-Collins 21 , Michael Nicolle 22 , Richard J Nowak 2 , Aditi Sharma 2 , Bhaskar Roy 2 , Joan Nye 2 , Michael Pulley 23 , Alan Berger 23 , Yasmeen Shabbir 23 , Amit Sachdev 24 , Kimberly Patterson 24 , Zaeem Siddiqi 25 , Mark Sivak 26 , Joan Bratton 26 , George Small 27 , Anem Kohli 27 , Mary Fetter 27 , Tuan Vu 5 , Lucy Lam 5 , Brittany Harvey 5 , Gil I Wolfe 3 , Nicholas Silvestri 3 , Kara Patrick 3 , Karen Zakalik 3 , Petra W Duda 8 , James MacDougall 8 , Ramin Farzaneh-Far 8 , Angela Pontius 8 , Michelle Hoarty 8
JAMA Neurology ( IF 29.0 ) Pub Date : 2020-05-01 , DOI: 10.1001/jamaneurol.2019.5125 James F Howard 1 , Richard J Nowak 2 , Gil I Wolfe 3 , Miriam L Freimer 4 , Tuan H Vu 5 , John L Hinton 6 , Michael Benatar 7 , Petra W Duda 8 , James E MacDougall 8 , Ramin Farzaneh-Far 8 , Henry J Kaminski 9 , , Richard Barohn 10 , Mazen Dimachkie 10 , Mamatha Pasnoor 10 , Constantine Farmakidis 10 , Tina Liu 10 , Samantha Colgan 10 , Michael G Benatar 7 , Tulio Bertorini 11 , Rekha Pillai 11 , Robert Henegar 11 , Mark Bromberg 12 , Summer Gibson 12 , Teresa Janecki 12 , Miriam Freimer 4 , Bakri Elsheikh 4 , Paige Matisak 4 , Angela Genge 13 , Amanda Guidon 14 , William David 14 , Ali A Habib 15 , Veena Mathew 15 , Tahseen Mozaffar 15 , John L Hinton 6 , William Hewitt 6 , Deborah Barnett 6 , Patricia Sullivan 6 , Doreen Ho 16 , James F Howard 1 , Rebecca E Traub 1 , Manisha Chopra 1 , Henry J Kaminski 9 , Radwa Aly 9 , Elham Bayat 9 , Mohammad Abu-Rub 9 , Shaida Khan 17 , Dale Lange 18 , Shara Holzberg 18 , Bhupendra Khatri 19 , Emily Lindman 19 , Tayo Olapo 19 , Lisa M Sershon 19 , Robert P Lisak 20 , Evanthia Bernitsas 20 , Kelly Jia 20 , Rabia Malik 21 , Tiffany D Lewis-Collins 21 , Michael Nicolle 22 , Richard J Nowak 2 , Aditi Sharma 2 , Bhaskar Roy 2 , Joan Nye 2 , Michael Pulley 23 , Alan Berger 23 , Yasmeen Shabbir 23 , Amit Sachdev 24 , Kimberly Patterson 24 , Zaeem Siddiqi 25 , Mark Sivak 26 , Joan Bratton 26 , George Small 27 , Anem Kohli 27 , Mary Fetter 27 , Tuan Vu 5 , Lucy Lam 5 , Brittany Harvey 5 , Gil I Wolfe 3 , Nicholas Silvestri 3 , Kara Patrick 3 , Karen Zakalik 3 , Petra W Duda 8 , James MacDougall 8 , Ramin Farzaneh-Far 8 , Angela Pontius 8 , Michelle Hoarty 8
Affiliation
|
Importance
Many patients with generalized myasthenia gravis (gMG) have substantial clinical disability, persistent disease burden, and adverse effects attributable to chronic immunosuppression. Therefore, there is a significant need for targeted, well-tolerated therapies with the potential to improve disease control and enhance quality of life.
Objective
To evaluate the clinical effects of zilucoplan, a subcutaneously (SC) self-administered macrocyclic peptide inhibitor of complement component 5, in a broad population of patients with moderate to severe gMG.
Design, Setting, and Participants
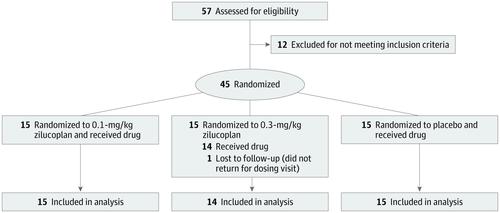
This randomized, double-blind, placebo-controlled phase 2 clinical trial at 25 study sites across North America recruited participants between December 2017 and August 2018. Fifty-seven patients were screened, of whom 12 did not meet inclusion criteria and 1 was lost to follow-up after randomization but before receiving study drug, resulting in a total of 44 acetylcholine receptor autoantibody (AChR-Ab)-positive patients with gMG with baseline Quantitative Myasthenia Gravis (QMG) scores of at least 12, regardless of treatment history.
Interventions
Patients were randomized 1:1:1 to a daily SC self-injection of placebo, 0.1-mg/kg zilucoplan, or 0.3-mg/kg zilucoplan for 12 weeks.
Main Outcomes and Measures
The primary and key secondary end points were the change from baseline to week 12 in QMG and MG Activities of Daily Living scores, respectively. Significance testing was prespecified at a 1-sided α of .10. Safety and tolerability were also assessed.
Results
The study of 44 patients was well balanced across the 3 treatment arms with respect to key demographic and disease-specific variables. The mean age of patients across all 3 treatment groups ranged from 45.5 to 54.6 years and most patients were white (average proportions across 3 treatment groups: 78.6%-86.7%). Clinically meaningful and statistically significant improvements in primary and key secondary efficacy end points were observed. Zilucoplan at a dose of 0.3 mg/kg SC daily resulted in a mean reduction from baseline of 6.0 points in the QMG score (placebo-corrected change, -2.8; P = .05) and 3.4 points in the MG Activities of Daily Living score (placebo-corrected change, -2.3; P = .04). Clinically meaningful and statistically significant improvements were also observed in other secondary end points, the MG Composite and MG Quality-of-Life scores. Outcomes for the 0.1-mg/kg SC daily dose were also statistically significant but slower in onset and less pronounced than with the 0.3-mg/kg dose. Rescue therapy (intravenous immunoglobulin or plasma exchange) was required in 3 of 15, 1 of 15, and 0 of 14 participants in the placebo, 0.1-mg/kg zilucoplan, and 0.3-mg/kg zilucoplan arms, respectively. Zilucoplan was observed to have a favorable safety and tolerability profile.
Conclusions and Relevance
Zilucoplan yielded rapid, meaningful, and sustained improvements over 12 weeks in a broad population of patients with moderate to severe AChR-Ab-positive gMG. Near-complete complement inhibition appeared superior to submaximal inhibition. The observed safety and tolerability profile of zilucoplan was favorable.
Trial Registration
ClinicalTrials.gov Identifier: NCT03315130.
中文翻译:

自我给药皮下补体抑制剂 Zilucoplan 对中重度全身性重症肌无力患者的临床效果:2 期随机、双盲、安慰剂对照、多中心临床试验的结果。
重要性 许多全身性重症肌无力 (gMG) 患者有严重的临床残疾、持续的疾病负担和慢性免疫抑制引起的不良反应。因此,迫切需要具有改善疾病控制和提高生活质量潜力的靶向性、耐受性良好的疗法。目的 评估 zilucoplan,一种皮下 (SC) 自我给药补体成分 5 的大环肽抑制剂,在广泛的中度至重度 gMG 患者中的临床效果。设计、设置和参与者这项在北美 25 个研究地点进行的随机、双盲、安慰剂对照的 2 期临床试验在 2017 年 12 月至 2018 年 8 月期间招募了参与者。 对 57 名患者进行了筛查,其中12人不符合纳入标准,1人在随机分组后但在接受研究药物之前失访,导致总共44例乙酰胆碱受体自身抗体(AChR-Ab)阳性gMG患者基线定量重症肌无力(QMG) ) 至少 12 分,无论治疗史如何。干预措施 患者按 1:1:1 随机接受每日皮下自我注射安慰剂、0.1-mg/kg zilucoplan 或 0.3-mg/kg zilucoplan,持续 12 周。主要结果和测量主要和关键次要终点分别是 QMG 和 MG 日常生活活动评分从基线到第 12 周的变化。显着性检验预先指定为 0.10 的单侧 α。还评估了安全性和耐受性。结果 44 名患者的研究在 3 个治疗组中在关键人口统计学和疾病特异性变量方面取得了很好的平衡。所有 3 个治疗组的患者平均年龄范围为 45.5 至 54.6 岁,大多数患者是白人(3 个治疗组的平均比例:78.6%-86.7%)。观察到主要和关键次要疗效终点具有临床意义和统计学意义的改善。每天 0.3 mg/kg SC 剂量的 Zilucoplan 导致 QMG 评分(安慰剂校正的变化,-2.8;P = .05)从基线平均降低 6.0 分,MG 日常生活活动评分降低 3.4 分(安慰剂校正的变化,-2.3;P = .04)。在其他次要终点中也观察到了具有临床意义和统计学意义的改善,MG 综合评分和 MG 生活质量评分。0.1-mg/kg SC 日剂量的结果也具有统计学意义,但起效较慢且不如 0.3-mg/kg 剂量明显。安慰剂组、0.1-mg/kg 齐卢普兰组和 0.3-mg/kg 齐卢普兰组分别有 3 位、15 位和 1 位和 14 位参与者需要抢救治疗(静脉注射免疫球蛋白或血浆置换)。观察到 Zilucoplan 具有良好的安全性和耐受性。结论和相关性 Zilucoplan 在 12 周内在广泛的中度至重度 AChR-Ab 阳性 gMG 患者群体中产生了快速、有意义和持续的改善。近乎完全的补体抑制似乎优于次最大抑制。观察到的 zilucoplan 的安全性和耐受性特征是有利的。试验注册临床试验。
更新日期:2020-05-01
中文翻译:

自我给药皮下补体抑制剂 Zilucoplan 对中重度全身性重症肌无力患者的临床效果:2 期随机、双盲、安慰剂对照、多中心临床试验的结果。
重要性 许多全身性重症肌无力 (gMG) 患者有严重的临床残疾、持续的疾病负担和慢性免疫抑制引起的不良反应。因此,迫切需要具有改善疾病控制和提高生活质量潜力的靶向性、耐受性良好的疗法。目的 评估 zilucoplan,一种皮下 (SC) 自我给药补体成分 5 的大环肽抑制剂,在广泛的中度至重度 gMG 患者中的临床效果。设计、设置和参与者这项在北美 25 个研究地点进行的随机、双盲、安慰剂对照的 2 期临床试验在 2017 年 12 月至 2018 年 8 月期间招募了参与者。 对 57 名患者进行了筛查,其中12人不符合纳入标准,1人在随机分组后但在接受研究药物之前失访,导致总共44例乙酰胆碱受体自身抗体(AChR-Ab)阳性gMG患者基线定量重症肌无力(QMG) ) 至少 12 分,无论治疗史如何。干预措施 患者按 1:1:1 随机接受每日皮下自我注射安慰剂、0.1-mg/kg zilucoplan 或 0.3-mg/kg zilucoplan,持续 12 周。主要结果和测量主要和关键次要终点分别是 QMG 和 MG 日常生活活动评分从基线到第 12 周的变化。显着性检验预先指定为 0.10 的单侧 α。还评估了安全性和耐受性。结果 44 名患者的研究在 3 个治疗组中在关键人口统计学和疾病特异性变量方面取得了很好的平衡。所有 3 个治疗组的患者平均年龄范围为 45.5 至 54.6 岁,大多数患者是白人(3 个治疗组的平均比例:78.6%-86.7%)。观察到主要和关键次要疗效终点具有临床意义和统计学意义的改善。每天 0.3 mg/kg SC 剂量的 Zilucoplan 导致 QMG 评分(安慰剂校正的变化,-2.8;P = .05)从基线平均降低 6.0 分,MG 日常生活活动评分降低 3.4 分(安慰剂校正的变化,-2.3;P = .04)。在其他次要终点中也观察到了具有临床意义和统计学意义的改善,MG 综合评分和 MG 生活质量评分。0.1-mg/kg SC 日剂量的结果也具有统计学意义,但起效较慢且不如 0.3-mg/kg 剂量明显。安慰剂组、0.1-mg/kg 齐卢普兰组和 0.3-mg/kg 齐卢普兰组分别有 3 位、15 位和 1 位和 14 位参与者需要抢救治疗(静脉注射免疫球蛋白或血浆置换)。观察到 Zilucoplan 具有良好的安全性和耐受性。结论和相关性 Zilucoplan 在 12 周内在广泛的中度至重度 AChR-Ab 阳性 gMG 患者群体中产生了快速、有意义和持续的改善。近乎完全的补体抑制似乎优于次最大抑制。观察到的 zilucoplan 的安全性和耐受性特征是有利的。试验注册临床试验。

























 京公网安备 11010802027423号
京公网安备 11010802027423号