当前位置:
X-MOL 学术
›
Nat. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Electronic complementarity permits hindered butenolide heterodimerization and discovery of novel cGAS/STING pathway antagonists.
Nature Chemistry ( IF 21.8 ) Pub Date : 2020-02-10 , DOI: 10.1038/s41557-019-0413-8 Benjamin J Huffman 1 , Shuming Chen 2 , J Luca Schwarz 1 , R Erik Plata 1 , Emily N Chin 1 , Luke L Lairson 1 , K N Houk 2 , Ryan A Shenvi 1
Nature Chemistry ( IF 21.8 ) Pub Date : 2020-02-10 , DOI: 10.1038/s41557-019-0413-8 Benjamin J Huffman 1 , Shuming Chen 2 , J Luca Schwarz 1 , R Erik Plata 1 , Emily N Chin 1 , Luke L Lairson 1 , K N Houk 2 , Ryan A Shenvi 1
Affiliation
|
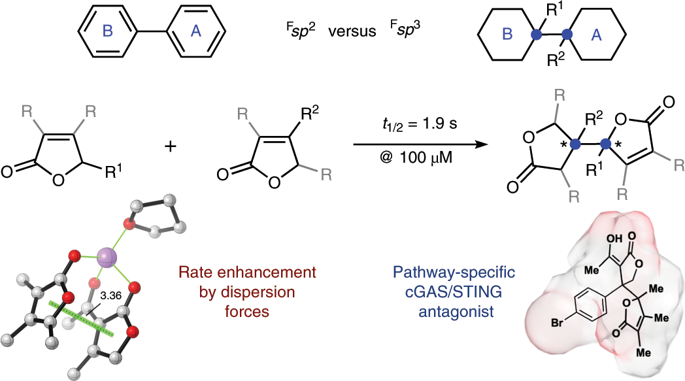
sp3-hybridized attached-rings are common motifs in secondary metabolites and represent tetrahedral equivalents to the biaryl substructures that overpopulate synthetic libraries. Few methods are available that can link fully substituted carbon atoms of two rings with stereocontrol. Here we have developed a stereoselective, heteroselective butenolide coupling that exhibits an unusually fast rate of C-C bond formation driven by exquisite complementarity of the reacting π systems. Heterodimerization generates a compound collection with topological complexity and diverse principal moments of inertia. The special status of the sp3-sp3 attached-ring motif is demonstrated in a high-throughput screen for inhibitors of the cyclic GMP-AMP synthase/stimulator of interferon genes pathway, which recruited these butenolide heterodimers from a field of 250,000 compounds. The driving forces underlying this general attached-ring coupling identify a novel paradigm for the accession of wider natural product chemical space, accelerating the discovery of selective lead compounds.
中文翻译:

电子互补性可阻止丁烯内酯异二聚化,并发现新型cGAS / STING途径拮抗剂。
sp3-杂化的连接环是次级代谢产物中的常见基序,代表合成库过剩的联芳基亚结构的四面体等价物。很少有方法可以通过立体控制连接两个环的完全取代的碳原子。在这里,我们开发了一种立体选择性,杂选择性丁烯内酯偶联剂,该偶联剂表现出由反应性π系统的精妙互补性驱动的CC键形成速率异常快的现象。异二聚化产生具有拓扑复杂性和各种主要惯性矩的化合物集合。在高通量筛选环GMP-AMP合酶/干扰素基因途径的抑制剂的过程中,sp3-sp3附着环基序的特殊状态得到了证明,该抑制剂从250个领域募集了这些丁烯内酯异二聚体,000种化合物。这种一般的连接环耦合的驱动力为进入更广阔的天然产物化学空间确定了一种新的范例,从而加速了选择性铅化合物的发现。
更新日期:2020-02-10
中文翻译:

电子互补性可阻止丁烯内酯异二聚化,并发现新型cGAS / STING途径拮抗剂。
sp3-杂化的连接环是次级代谢产物中的常见基序,代表合成库过剩的联芳基亚结构的四面体等价物。很少有方法可以通过立体控制连接两个环的完全取代的碳原子。在这里,我们开发了一种立体选择性,杂选择性丁烯内酯偶联剂,该偶联剂表现出由反应性π系统的精妙互补性驱动的CC键形成速率异常快的现象。异二聚化产生具有拓扑复杂性和各种主要惯性矩的化合物集合。在高通量筛选环GMP-AMP合酶/干扰素基因途径的抑制剂的过程中,sp3-sp3附着环基序的特殊状态得到了证明,该抑制剂从250个领域募集了这些丁烯内酯异二聚体,000种化合物。这种一般的连接环耦合的驱动力为进入更广阔的天然产物化学空间确定了一种新的范例,从而加速了选择性铅化合物的发现。

























 京公网安备 11010802027423号
京公网安备 11010802027423号