Catalysis Communications ( IF 3.7 ) Pub Date : 2020-01-21 , DOI: 10.1016/j.catcom.2020.105942 Marzieh Rafiee , Hadis Bashiri
|
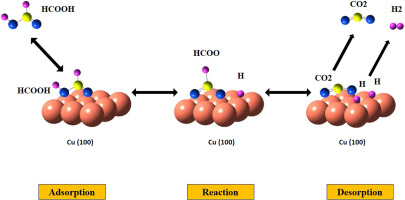
Dynamic Monte Carlo simulation and response surface methodology have been used to study optimum conditions for the heterogeneous catalytic formic acid decomposition reaction on Cu(100) for the first time. The mechanism, kinetic parameters, optimum conditions and the effects of temperature, pressure and reaction time on the hydrogen yield were studied.. Formate (HCOO) species is the active reaction intermediate and the adsorption of formic acid is considered as the rate-controlling step. The proposed optimum conditions provided a turnover frequency (TOF) of 0.044 s−1 for hydrogen production.
中文翻译:

Cu(100)上甲酸的催化分解:优化和动态蒙特卡洛模拟
动态蒙特卡罗模拟和响应表面方法已被用于研究最佳条件的铜(100)上的非均相催化甲酸分解反应。研究了机理,动力学参数,最佳条件以及温度,压力和反应时间对氢气产率的影响。甲酸酯类(HCOO)是活性反应中间体,甲酸的吸附被认为是控制速率的步骤。提出的最佳条件为制氢提供了0.044 s -1的周转频率(TOF)。


























 京公网安备 11010802027423号
京公网安备 11010802027423号