Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Heat Shock Factor 2 Protects against Proteotoxicity by Maintaining Cell-Cell Adhesion.
Cell Reports ( IF 8.8 ) Pub Date : 2020-01-14 , DOI: 10.1016/j.celrep.2019.12.037 Jenny Joutsen 1 , Alejandro Jose Da Silva 1 , Jens Christian Luoto 1 , Marek Andrzej Budzynski 1 , Anna Serafia Nylund 1 , Aurelie de Thonel 2 , Jean-Paul Concordet 3 , Valérie Mezger 2 , Délara Sabéran-Djoneidi 2 , Eva Henriksson 1 , Lea Sistonen 1
Cell Reports ( IF 8.8 ) Pub Date : 2020-01-14 , DOI: 10.1016/j.celrep.2019.12.037 Jenny Joutsen 1 , Alejandro Jose Da Silva 1 , Jens Christian Luoto 1 , Marek Andrzej Budzynski 1 , Anna Serafia Nylund 1 , Aurelie de Thonel 2 , Jean-Paul Concordet 3 , Valérie Mezger 2 , Délara Sabéran-Djoneidi 2 , Eva Henriksson 1 , Lea Sistonen 1
Affiliation
|
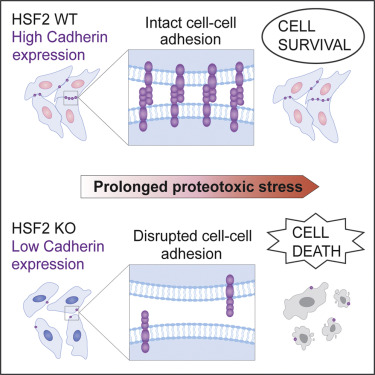
Maintenance of protein homeostasis, through inducible expression of molecular chaperones, is essential for cell survival under protein-damaging conditions. The expression and DNA-binding activity of heat shock factor 2 (HSF2), a member of the heat shock transcription factor family, increase upon exposure to prolonged proteotoxicity. Nevertheless, the specific roles of HSF2 and the global HSF2-dependent gene expression profile during sustained stress have remained unknown. Here, we found that HSF2 is critical for cell survival during prolonged proteotoxicity. Strikingly, our RNA sequencing (RNA-seq) analyses revealed that impaired viability of HSF2-deficient cells is not caused by inadequate induction of molecular chaperones but is due to marked downregulation of cadherin superfamily genes. We demonstrate that HSF2-dependent maintenance of cadherin-mediated cell-cell adhesion is required for protection against stress induced by proteasome inhibition. This study identifies HSF2 as a key regulator of cadherin superfamily genes and defines cell-cell adhesion as a determinant of proteotoxic stress resistance.
中文翻译:

热激因子2通过维持细胞间粘附力来防止蛋白毒性。
通过分子伴侣的可诱导表达来维持蛋白质稳态,对于蛋白质破坏条件下的细胞存活至关重要。热休克因子2(HSF2)(热休克转录因子家族的成员)的表达和DNA结合活性在长期暴露于蛋白毒性时会增加。然而,在持续的应激过程中,HSF2的具体作用和整体HSF2依赖性基因表达谱仍然未知。在这里,我们发现HSF2对于长时间蛋白毒性期间的细胞存活至关重要。令人惊讶的是,我们的RNA测序(RNA-seq)分析表明,HSF2缺陷型细胞的活力受损并非由分子伴侣的诱导不足引起,而是由于钙粘蛋白超家族基因的显着下调。我们证明,HSF2依赖钙黏着蛋白介导的细胞粘附的维持对于保护抵抗蛋白酶体抑制诱导的应激是必需的。这项研究确定了HSF2是钙粘蛋白超家族基因的关键调节因子,并将细胞粘附确定为蛋白毒性应激抗性的决定因素。
更新日期:2020-01-15
中文翻译:

热激因子2通过维持细胞间粘附力来防止蛋白毒性。
通过分子伴侣的可诱导表达来维持蛋白质稳态,对于蛋白质破坏条件下的细胞存活至关重要。热休克因子2(HSF2)(热休克转录因子家族的成员)的表达和DNA结合活性在长期暴露于蛋白毒性时会增加。然而,在持续的应激过程中,HSF2的具体作用和整体HSF2依赖性基因表达谱仍然未知。在这里,我们发现HSF2对于长时间蛋白毒性期间的细胞存活至关重要。令人惊讶的是,我们的RNA测序(RNA-seq)分析表明,HSF2缺陷型细胞的活力受损并非由分子伴侣的诱导不足引起,而是由于钙粘蛋白超家族基因的显着下调。我们证明,HSF2依赖钙黏着蛋白介导的细胞粘附的维持对于保护抵抗蛋白酶体抑制诱导的应激是必需的。这项研究确定了HSF2是钙粘蛋白超家族基因的关键调节因子,并将细胞粘附确定为蛋白毒性应激抗性的决定因素。

























 京公网安备 11010802027423号
京公网安备 11010802027423号