当前位置:
X-MOL 学术
›
Acta Biomater.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Tumor-targeted nanoplatform for in situ oxygenation-boosted immunogenic phototherapy of colorectal cancer.
Acta Biomaterialia ( IF 9.7 ) Pub Date : 2020-01-13 , DOI: 10.1016/j.actbio.2020.01.012 Huamei He 1 , Lanlan Liu 2 , Ruijing Liang 1 , Haimei Zhou 1 , Hong Pan 1 , Shengping Zhang 1 , Lintao Cai 1
Acta Biomaterialia ( IF 9.7 ) Pub Date : 2020-01-13 , DOI: 10.1016/j.actbio.2020.01.012 Huamei He 1 , Lanlan Liu 2 , Ruijing Liang 1 , Haimei Zhou 1 , Hong Pan 1 , Shengping Zhang 1 , Lintao Cai 1
Affiliation
|
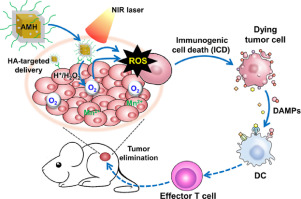
Advanced colorectal cancer has a high mortality rate since conventional treatments have limited therapeutic effects and poor prognosis with high risks of metastasis and recurrence. Photodynamic therapy (PDT) is a promising treatment modality for the eradication of colorectal cancer, but its curative efficacy is severely affected by tumor hypoxia. Herein, we developed a core-shell gold nanocage coated with manganese dioxide and hyaluronic acid (AMH) for targeted delivery to colorectal tumors and oxygenation-boosted immunogenic phototherapy in situ. The AMH nanoparticles can generate abundant oxygen from mild acidic/H2O2 medium, which can further enhance the PDT efficacy of AMH itself under near infrared (NIR) irradiation. Meanwhile, AMH-based PDT induced immunogenic cell death (ICD) of tumor cells with damage-associated molecular patterns (DAMPs) release and facilitated the dendritic cells (DCs) maturation to further potentiate the systematic antitumor immunity against advanced tumors. In vivo experiment results exhibited that AMH nanoparticles not only had the ability of targeting tumor but also in situ produced sufficient oxygen to relieve the tumor hypoxia. Furthermore, AMH-mediated oxygen-boosted immunogenic PDT effectively inhibited the tumor growth and recurrence. Thus, this work provides a potent targeted delivery nanoplatform for enhanced immunogenic PDT against advanced cancers. STATEMENT OF SIGNIFICANCE: Local hypoxic tumor microenvironment not only greatly limits the photodynamic therapy (PDT) efficacy, but also has an association with tumor invasiveness and metastasis. This study provides an AMH nanoparticle for targeted delivery to colorectal tumors and oxygenation-boosted immunogenic PDT in situ. AMH nanoparticle exhibits a good tumor-targeted ability to in situ produce abundant oxygen to relieve the tumor hypoxia, and initiates the potent oxygen-boosted immunogenic PDT effect under NIR irradiation to effectively inhibit the growth and recurrence of colorectal tumor. This oxygen-boosted immunogenic PDT nanosystem can be a promising candidate for advanced tumor treatment.
中文翻译:

针对大肠癌的原位充氧增强免疫原性光疗的肿瘤靶向纳米平台。
晚期结直肠癌的死亡率很高,因为常规治疗的疗效有限且预后较差,且转移和复发的风险较高。光动力疗法(PDT)是根除大肠癌的一种有前途的治疗方法,但其疗效受到肿瘤缺氧的严重影响。在本文中,我们开发了包覆有二氧化锰和透明质酸(AMH)的核壳金纳米笼,用于靶向递送至结直肠肿瘤和氧合作用原位免疫原性光疗。AMH纳米颗粒可以从弱酸性/ H2O2介质中产生大量氧气,这可以进一步增强AMH自身在近红外(NIR)辐射下的PDT功效。与此同时,基于AMH的PDT诱导具有损伤相关分子模式(DAMP)的肿瘤细胞的免疫原性细胞死亡(ICD)释放,并促进树突状细胞(DC)成熟,从而进一步增强了针对晚期肿瘤的系统性抗肿瘤免疫力。体内实验结果表明,AMH纳米粒子不仅具有靶向肿瘤的能力,而且还就地产生足够的氧气以缓解肿瘤的缺氧。此外,AMH介导的增氧免疫原性PDT有效抑制了肿瘤的生长和复发。因此,这项工作为增强的针对晚期癌症的免疫原性PDT提供了有效的靶向递送纳米平台。重要性声明:局部低氧肿瘤微环境不仅极大地限制了光动力疗法(PDT)的功效,但也与肿瘤的侵袭和转移有关。这项研究提供了一种AMH纳米颗粒,用于靶向递送至大肠肿瘤和原位充氧增强免疫原性PDT。AMH纳米颗粒具有良好的针对肿瘤的原位产生大量氧气的能力,以缓解肿瘤的缺氧,并在NIR照射下启动了强大的增氧免疫原性PDT效应,从而有效抑制了结直肠肿瘤的生长和复发。这种增氧的免疫原性PDT纳米系统可以成为晚期肿瘤治疗的有希望的候选者。并在NIR照射下启动强大的增氧免疫原性PDT效应,以有效抑制结直肠肿瘤的生长和复发。这种增氧的免疫原性PDT纳米系统可以成为晚期肿瘤治疗的有希望的候选者。并在NIR照射下启动强大的增氧免疫原性PDT效应,以有效抑制结直肠肿瘤的生长和复发。这种增氧的免疫原性PDT纳米系统可以成为晚期肿瘤治疗的有希望的候选者。
更新日期:2020-01-13
中文翻译:

针对大肠癌的原位充氧增强免疫原性光疗的肿瘤靶向纳米平台。
晚期结直肠癌的死亡率很高,因为常规治疗的疗效有限且预后较差,且转移和复发的风险较高。光动力疗法(PDT)是根除大肠癌的一种有前途的治疗方法,但其疗效受到肿瘤缺氧的严重影响。在本文中,我们开发了包覆有二氧化锰和透明质酸(AMH)的核壳金纳米笼,用于靶向递送至结直肠肿瘤和氧合作用原位免疫原性光疗。AMH纳米颗粒可以从弱酸性/ H2O2介质中产生大量氧气,这可以进一步增强AMH自身在近红外(NIR)辐射下的PDT功效。与此同时,基于AMH的PDT诱导具有损伤相关分子模式(DAMP)的肿瘤细胞的免疫原性细胞死亡(ICD)释放,并促进树突状细胞(DC)成熟,从而进一步增强了针对晚期肿瘤的系统性抗肿瘤免疫力。体内实验结果表明,AMH纳米粒子不仅具有靶向肿瘤的能力,而且还就地产生足够的氧气以缓解肿瘤的缺氧。此外,AMH介导的增氧免疫原性PDT有效抑制了肿瘤的生长和复发。因此,这项工作为增强的针对晚期癌症的免疫原性PDT提供了有效的靶向递送纳米平台。重要性声明:局部低氧肿瘤微环境不仅极大地限制了光动力疗法(PDT)的功效,但也与肿瘤的侵袭和转移有关。这项研究提供了一种AMH纳米颗粒,用于靶向递送至大肠肿瘤和原位充氧增强免疫原性PDT。AMH纳米颗粒具有良好的针对肿瘤的原位产生大量氧气的能力,以缓解肿瘤的缺氧,并在NIR照射下启动了强大的增氧免疫原性PDT效应,从而有效抑制了结直肠肿瘤的生长和复发。这种增氧的免疫原性PDT纳米系统可以成为晚期肿瘤治疗的有希望的候选者。并在NIR照射下启动强大的增氧免疫原性PDT效应,以有效抑制结直肠肿瘤的生长和复发。这种增氧的免疫原性PDT纳米系统可以成为晚期肿瘤治疗的有希望的候选者。并在NIR照射下启动强大的增氧免疫原性PDT效应,以有效抑制结直肠肿瘤的生长和复发。这种增氧的免疫原性PDT纳米系统可以成为晚期肿瘤治疗的有希望的候选者。

























 京公网安备 11010802027423号
京公网安备 11010802027423号