Current Computer-Aided Drug Design ( IF 1.7 ) Pub Date : 2020-09-30 , DOI: 10.2174/1573409915666190902143648 Devidas G Anuse 1 , Bapu R Thorat 1 , Sudhir Sawant 1 , Ramesh S Yamgar 2 , Hemchandra K Chaudhari 3 , Suraj N Mali 3
|
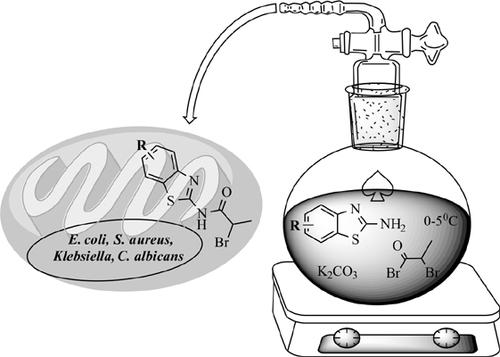
Background: Benzothiazoles are reported to have bioorganic and pharmaceutical chemistry applications.
Introduction: A series of substituted N-bromoamido-2-aminobenzothiazoles was synthesized from substituted anilines via 2-aminobenzothiazoles and it was further evaluated for its antimicrobial activity.
Methods: All the newly synthesized compounds were characterized by FT-IR, NMR and mass spectra and purity profiles were studied by HPLC analysis. The antimicrobial testing (MIC determination) was newly performed with agar micro-broth dilution method for these analogs.
Results: Among the synthesized compound 3b showed the highest activity with MIC value of 3.12 μg/mL against Bacillus, E. coli, S. aureus and Klebsiella and 6.25 μg/mL against C. albicans. The ADME properties as calculated by using Qikprop were found within acceptable range. Derivatives shows a good-moderate binding affinity towards target Cytochrome P450 14 alpha-sterol demethylase (CYP51) (PDB ID: 1EA1).
Conclusion: Our in-silico and in-vitro studies on a series of substituted aminobenzothiazoles may be helpful for further designing of more potent antimicrobials in future.
中文翻译:

取代的 N-溴酰胺-2-氨基苯并噻唑的合成、合成孔径雷达、分子对接和抗微生物研究。
背景:据报道苯并噻唑具有生物有机和药物化学应用。
简介:以取代苯胺为原料,通过2-氨基苯并噻唑合成了一系列取代的N-溴酰胺-2-氨基苯并噻唑,并对其抗菌活性进行了进一步评价。
方法:所有新合成的化合物均通过 FT-IR、NMR 和质谱进行表征,并通过 HPLC 分析研究纯度曲线。对这些类似物使用琼脂微量肉汤稀释法新进行了抗菌测试(MIC 测定)。
结果:合成的化合物中3b活性最高,对芽孢杆菌、大肠杆菌、金黄色葡萄球菌和克雷伯菌的MIC值为3.12 μg/mL,对白色念珠菌的MIC值为6.25 μg/mL。发现使用 Qikprop 计算的 ADME 属性在可接受的范围内。衍生物对目标细胞色素 P450 14 α-甾醇脱甲基酶 (CYP51) (PDB ID: 1EA1) 显示出良好的中等结合亲和力。
结论:我们对一系列取代氨基苯并噻唑的计算机内和体外研究可能有助于未来进一步设计更有效的抗菌剂。


























 京公网安备 11010802027423号
京公网安备 11010802027423号