RSC主编推荐:有机领域精彩文章快览(免费阅读原文)
英国皇家化学会(RSC)是一个超过175年历史的面向全球化学家的非营利会员制机构,旗下拥有44种期刊,其中很多在化学领域有很高影响力。为了进一步帮助广大读者追踪科技前沿热点,X-MOL团队与英国皇家化学会合作,推出英国皇家化学会期刊主编推荐的精彩文章快览,本期文章属“有机领域”,英文点评来自英国皇家化学会期刊的主编。如果大家对我们的解读有更多的补充和点评,欢迎在文末写评论发表您的高见!
Chemical Science (IF: 9.063)

1. Hydroxy Group-Enabled Highly Regio- and Stereo-selective Hydrocarboxylation of Alkynes
Chem. Sci., 2019, Advance Article
DOI: 10.1039/C8SC05743E
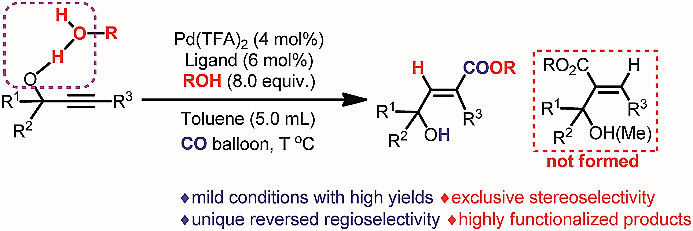
Here the authors present a highly efficient Pd-catalyzed syn-hydrocarboxylation of readily available 2-alkynylic alcohols with CO in the presence of alcohols with an unprecedented regioselectivity affording 3-hydroxy-2(E)-alkenoates. This shows an example of being able to utilise hydroxy groups for regioselectivity control in the addition reaction of alkynes.
作者报道了在醇的存在下易获取的2-炔基醇与一氧化碳的高效钯催化syn-氢羧化反应,以高区域选择性制备了3-羟基-2(E)-烯酸酯。这为利用羟基控制炔加成反应的区域选择性提供了实例。
Open Access(可免费阅读原文)
扫描或长按二维码,识别后直达原文页面,或点此查看原文

2. Exploiting the Trifluoroethyl Group as a Precatalyst Ligand in Nickel-catalyzed Suzuki-type Alkylations
Chem. Sci., 2019, Advance Article
DOI: 10.1039/C9SC00554D

The authors demonstrate here the utilisation of a partially fluorinated trifluoroethyl as a precatalyst ligand in nickel-catalyzed Suzuki-type alkylation and fluoroalkylation coupling reactions. Compared with the [LnNiII(aryl)(X)] precatalysts, the unique characteristics of the bis-trifluoroethyl ligands helped to prepare precatalysts [(bipy)Ni(CH2CF3)2] with bench-top stability, good solubility in organic media and interesting catalytic activities.
作者报道部分氟化的三氟乙基作为预催化剂配体,可用于镍催化的Sukuzi型烷基化和氟烷基化偶联反应。与[LnNiII(aryl)(X)]预催化剂相比,双三氟乙基配体独特的性质有助于制备预催化剂[(bipy)Ni(CH2CF3)2],该预催化剂具有实验室环境稳定性、良好的有机介质溶解性和令人关注的催化活性。
Open Access(可免费阅读原文)
扫描或长按二维码,识别后直达原文页面,或点此查看原文

Organic Chemistry Frontiers (IF: 5.455)

1. Quantification of the hydrophobic effect using water-soluble super aryl-extended calix[4]pyrroles
Org. Chem. Front., 2019, Advance Article
DOI: 10.1039/C9QO00171A
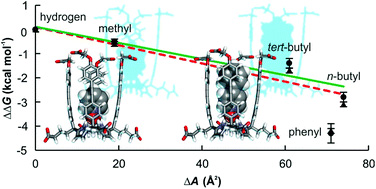
We describe the syntheses of the tetra-α isomers of three unprecedented super aryl-extended calix[4]pyrroles (SAE-C[4]Ps) functionalized with either eight carboxylic acids, eight pyridinium ions or eight 1-methyl-1H-imidazolium ions. We report the results of the binding studies of a series of pyridyl N-oxides, having a non-polar para-substituent, with the SAE-C[4]P receptors in water using 1H NMR spectroscopy titrations and isothermal titration calorimetry (ITC) experiments.
本文作者介绍了三种前所未有的超芳基扩展的杯[4]吡咯(SAE-C[4]P)的四-α异构体的合成,该杯[4]吡咯带有八个羧酸、吡啶离子或1-甲基-1H-咪唑鎓离子官能团;并使用1H NMR光谱滴定和等温滴定量热法(ITC)研究了一系列具有非极性对位取代基的吡啶基N-氧化物与SAE-C[4]P受体在水中的结合。
限时免费阅读原文,登录后可下载
扫描或长按二维码,识别后直达原文页面,或点此查看原文

2. Palladium-catalyzed dearomative arylphosphorylation of indoles
Org. Chem. Front., 2019, Advance Article
DOI: 10.1039/C9QO00246D
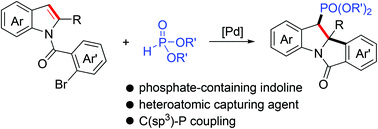
A palladium-catalyzed dearomative arylphosphorylation reaction of indoles involving C–C/C–P bond formation is developed. Through the intramolecular dearomative carbopalladation of indoles followed by an intermolecular benzyl–Pd termination with dialkylphosphite, a range of unique indolines bearing vicinal tetrasubstituted and phosphate-containing trisubstituted stereocenters are obtained in good yields. Synthetic transformations of the product show practical utilities of the reaction.
本文报道了一种涉及C-C/C-P键形成的钯催化的吲哚去芳构化芳基磷酸化反应。通过吲哚的分子内去芳构化碳钯化,以及随后的与亚磷酸二烷基酯的分子间苄基-钯终止反应,以良好产率制备了一系列包括相邻的四取代和含磷酸酯基团三取代立体中心的独特吲哚啉类化合物。产物的合成转化证明了该反应的实际应用潜力。
限时免费阅读原文,登录后可下载
扫描或长按二维码,识别后直达原文页面,或点此查看原文

如果篇首注明了授权来源,任何转载需获得来源方的许可!如果篇首未特别注明出处,本文版权属于 X-MOL ( x-mol.com ), 未经许可,谢绝转载!




























 京公网安备 11010802027423号
京公网安备 11010802027423号